Gwangju Institute of Science and Technology (GIST) announced on the 3rd that the research team led by Professor Jeongwook Kim of the Department of Chemistry has identified a new enzyme reaction mechanism that directly utilizes oxygen in the process by which living organisms accurately produce proteins.
This study is the first to explain, at the molecular level, how cells prevent errors during protein synthesis, and at the same time, it is the first achievement to reveal that an enzyme can directly utilize oxygen even without complex auxiliary factors such as metal ions or organic cofactors.
Through this, the research expands our understanding of existing enzyme reactions and presents the possibility of designing new biocatalysts that can achieve low-cost and eco-friendly hydroxylation reactions (reactions that change the properties of a molecule by attaching an oxygen component to it) in the future.
The genetic information in DNA, which serves as the blueprint of living organisms, is transferred to RNA and then translated into proteins through the translation process. Proteins form the structure of cells and are key substances that carry out vital activities, and even a single error can lead to cellular dysfunction or disease.
tRNA (transfer RNA) plays an important role in preventing such errors. tRNA accurately reads codons, which are genetic codes, and delivers amino acids, the basic building blocks of proteins, thereby helping proteins to be synthesized in the correct order.
To increase the accuracy of protein synthesis, cells undergo a process called "tRNA modification," in which specific components (bases) of tRNA are chemically altered. In particular, the wobble position of tRNA is a key site that allows a single tRNA to recognize multiple codons, and subtle chemical modifications at this position play a decisive role in maintaining translational fidelity.
The TrhO enzyme introduces a hydroxyl group into the uridine base located at the wobble position of tRNA, thereby helping ensure that genetic codes are decoded accurately. However, how TrhO activates molecular oxygen and delivers that oxygen to the target uridine had remained unknown.
In general, enzymes that use oxygen rely on the help of metal ions such as iron or copper, or organic cofactors such as FAD and NAD, but it was not known whether TrhO uses such cofactors in its reaction. As a result, the reaction principle of TrhO had remained an important unsolved problem.
To elucidate this reaction principle, it is important to determine what structure the enzyme adopts when it is actually bound to its reaction substrate. However, the TrhO structures reported so far have corresponded to states in which no reaction occurs, so it was not possible to understand how the enzyme operates when bound to tRNA.
In particular, this enzyme is smaller than TrhO enzymes found in other organisms, which has limited the interpretation of how it recognizes and modifies its reaction substrate.
To overcome these limitations, the research team used cryogenic electron microscopy (cryo-EM)* to determine, for the first time in the world, the structure of the TrhO enzyme in its active state while bound to tRNA. Through this, they were able to observe for the first time how TrhO recognizes tRNA and how a partial structural rearrangement of tRNA positions the uridine at the reaction site precisely at the catalytic center of the enzyme.
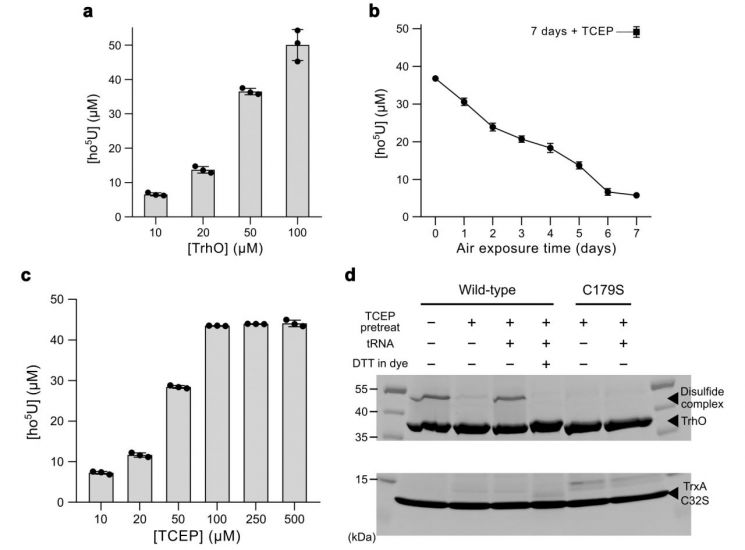
Analysis of the TrhO-tRNA complex structure revealed that the uridine at a key position in tRNA is accurately placed at the catalytic center of the TrhO enzyme, where it was observed to interact closely with a cysteine amino acid residue that directly drives the reaction. This provides structural evidence that this cysteine plays a key catalytic role in initiating the oxygen-based hydroxylation reaction.
Subsequent experiments confirmed that the TrhO enzyme can carry out this hydroxylation reaction even in the absence of metal ions or separate auxiliary factors. In fact, analysis of the purified enzyme detected no organic cofactors that assist the reaction, and the zinc ion observed in the structure was also found not to affect the enzyme's activity.
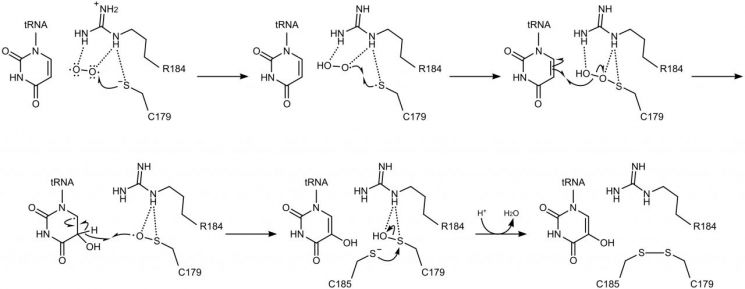
Based on these results, the research team proposed a completely new hydroxylation reaction mechanism in which the cysteine residue reacts directly with molecular oxygen, briefly forming an intermediate state and then transferring the oxygen component to the uridine located at the wobble position. This is fundamentally different from existing oxygen-utilizing enzymes that depend on metal ions or separate auxiliary factors.
Professor Jeongwook Kim stated, "This study has elucidated the principle by which a single enzyme alone, without the help of other auxiliary substances, can use molecular oxygen to introduce oxygen into a chemically very unreactive structure (an 'aromatic ring')," and added, "By harnessing the properties of this enzyme, we expect to simplify existing complex and costly chemical processes, achieve low-cost and highly efficient hydroxylation reactions, and eventually extend this work into technologies for designing and engineering practically useful enzymes."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.