Insurance Coverage Standards for Anticancer Drugs Revised from May
Patient Groups Welcome: "No More Abandoning Treatment for Financial Reasons"
As it is now possible to receive health insurance benefits for existing anticancer drugs even when used in combination with newly developed, non-reimbursed anticancer medications, cancer patients are expected to have improved access to treatment. Pharmaceutical companies are also expected to accelerate additional research and development, as they can now utilize a wider range of new drugs in cancer therapy.
According to the Ministry of Health and Welfare and the pharmaceutical industry on April 24, the recently announced administrative notice on the "Detailed Standards and Methods for the Application of Medical Care Benefits (Drugs) Partial Amendment" will take effect from May 1. The amendment stipulates that when a new anticancer drug approved by the Ministry of Food and Drug Safety is used in combination with an existing anticancer drug already covered by health insurance, the patient’s out-of-pocket payment for the existing reimbursed drug will remain unchanged.
Previously, when combination therapy involved both an insured anticancer drug and an expensive, newly introduced non-reimbursed drug, the entire treatment cost was excluded from insurance coverage. As a result, there were frequent cases where patients discontinued or gave up treatment for financial reasons.
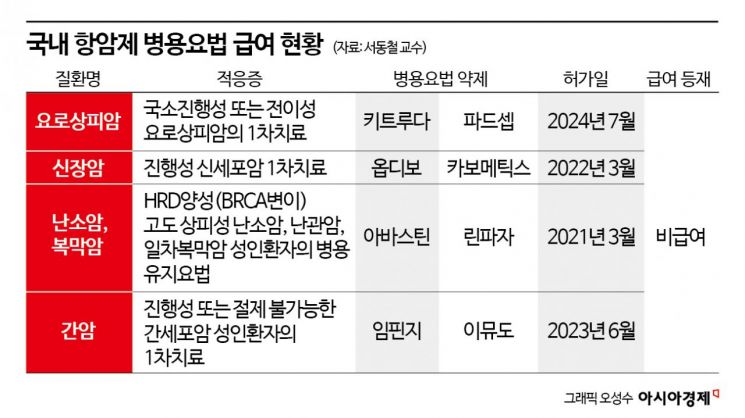
However, in recent cancer treatment, combination therapy?using two or more drugs together to enhance therapeutic effects?has become the new standard, rather than relying on a single anticancer drug alone. Over the past ten years, there have been 71 cases of combination anticancer therapies approved by the Ministry of Food and Drug Safety, with more than 75% (54 cases) concentrated in the past five years, indicating a rapid adoption of combination therapies. Among the combination therapies approved in the past five years, 28 cases involved combining existing anticancer drugs with new drugs, and 26 cases involved combining two new drugs, each accounting for half of the total. In particular, combination therapies involving immunotherapy and targeted therapy are actively being researched and developed, as they are expected to improve patient survival rates and reduce side effects simultaneously; however, all such therapies remain non-reimbursed in Korea.
Patients have evaluated the Ministry’s decision as a significant advancement that greatly improves access to pharmaceuticals and corrects long-standing unreasonable health insurance standards. The Korea Alliance of Patient Organizations stated, "When patients used non-reimbursed new drugs together with existing reimbursed drugs, they could not receive insurance benefits for the existing drugs either, leaving them to bear the full cost of treatment." The organization added, "Given that it typically takes more than a year for a new drug to be listed for health insurance coverage, this measure is expected to significantly help prevent situations where patients abandon treatment for financial reasons."
The Korea Research-based Pharmaceutical Industry Association (KRPIA), led by multinational pharmaceutical companies, also commented, "This amendment will substantially ease the financial burden of cancer patients, including treatment costs, while providing opportunities to choose innovative and effective therapies for survival." The association added, "We will continue to work toward the development of innovative new drugs and the creation of a patient-centered treatment environment."
However, while the path has been opened for reimbursement of combination anticancer therapies?a long-standing wish of cancer patients?it is expected to take considerable time before combination therapies involving new drugs and new drugs, which are emerging as a new paradigm in cancer treatment, are also covered by insurance. This is because, although cancer therapies are advancing rapidly, domestic systems related to new drug and combination therapy approvals and reimbursement are still unable to keep pace. In Korea, only 22% of global new drugs are covered by health insurance, and it takes an average of 46 months from initial launch to reimbursement.
From the government’s perspective, if all rapidly increasing combination anticancer therapies were to be reimbursed, it could place a burden on the health insurance budget. In addition, combination therapies often involve expensive drugs from different pharmaceutical companies, and each company may not be proactive in price negotiations during the reimbursement process, considering their respective R&D investments and profitability.
Seo Dongcheol, Professor Emeritus at Chung-Ang University College of Pharmacy, pointed out, "If the current criteria, which focus on cost-effectiveness (ICER) in economic evaluations rather than considering the severity of the disease, social needs, or patients’ quality of life during the processes of new drug approval, drug pricing, and reimbursement decisions, are not improved, the introduction of new drugs may be delayed and there may be difficulties in allocating drug costs when introducing combination therapies."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















