Korea-Japan Joint Research:
"Rapid Hydrogen Movement in Solids, Like in Liquids"
 (From left) Professor Kim Sangryun, integrated master's and doctoral students Kim Taehyun, Kim Taeseung, and Lee Taekyung
(From left) Professor Kim Sangryun, integrated master's and doctoral students Kim Taehyun, Kim Taeseung, and Lee Taekyung
A new technology that enables hydrogen to exist in an anionic state and move rapidly through solids, much like in a liquid, has been developed through an international joint research effort between Korea and Japan.
On April 21, the Gwangju Institute of Science and Technology (GIST) announced that a research team led by Professor Kim Sangryun of the Department of Chemistry (also affiliated with the School of Energy Convergence) has, in collaboration with the Korea Atomic Energy Research Institute and the Institute of Science Tokyo in Japan, demonstrated that it is possible to dramatically increase the conduction speed of hydrogen anions within a solid.
This research introduces an energy transfer technology fundamentally different from conventional lithium-ion or all-solid-state batteries. It significantly enhances the feasibility of eco-friendly secondary batteries, fuel cells, and water electrolysis technologies that utilize hydrogen anions, thereby holding high academic and industrial value.
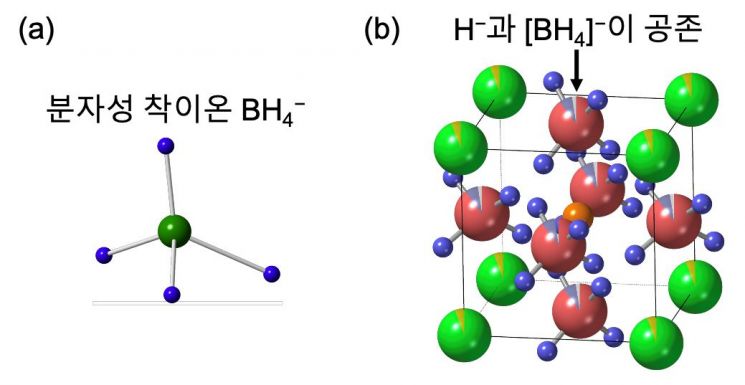
The study utilized paramagnetic complex anions. Complex anions are polyatomic ions with a central atom surrounded by multiple molecules or ions, carrying an electric charge. In this research, borohydride anions were used.
Generally, ions have difficulty moving freely in a solid state. However, if specific ions can move rapidly within a solid, it could bring about groundbreaking advancements in energy storage and conversion technologies, such as lithium-ion all-solid-state batteries. For this reason, the discovery of new conductive ions is considered a highly significant achievement not only academically but also in terms of future industrial applications.
Hydrogen is attracting attention as an eco-friendly energy source. However, it is difficult to handle stably due to its tendency to react easily with other substances. To address this, the research team designed a new structure that stabilizes hydrogen in an anionic form and enables its rapid movement within a solid, utilizing paramagnetic complex anions.
The research team discovered that borohydride anions, which possess strong electron-donating properties (high reducibility), can stabilize hydrogen in an anionic state. Based on this finding, they succeeded in synthesizing a new single-phase (homogeneous crystal structure) material in which borohydride anions and hydrogen anions coexist.
Additionally, through Maximum Entropy Method (MEM) analysis, the team found that the complex anions interact asymmetrically with surrounding ions and are fixed in place. Of particular note, hydrogen anions moved much more rapidly along pathways where the electrostatic interactions created by these asymmetric complex anions were weaker. In fact, compared to conventional structures without complex anions, the ionic conductivity of hydrogen anions increased by more than 1,000 times.
Professor Kim Sangryun stated, "This research is the world's first case of hydrogen anion conduction enabled by complex anions," adding, "It will serve as an important turning point for the future development of new eco-friendly hydrogen-based energy technologies."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.