A technology has been developed that enhances the performance of lithium metal batteries, enabling electric vehicles to travel up to 900 km on a single charge.
KAIST announced on the 7th that Professor Hee-Tak Kim from the Department of Bio and Chemical Engineering and a joint research team with LG Energy Solution have developed a core technology that can dramatically increase the performance of lithium metal batteries, which are attracting attention as next-generation batteries.
 (From left) Professor Kim Hee-tak, Department of Biological Chemical Engineering, and PhD candidate Kwon Hyuk-jin, Department of Biological Chemical Engineering. Provided by KAIST
(From left) Professor Kim Hee-tak, Department of Biological Chemical Engineering, and PhD candidate Kwon Hyuk-jin, Department of Biological Chemical Engineering. Provided by KAIST
The technology developed by the joint research team is significant in that it allows electric vehicles to travel up to 900 km on a single charge of a lithium metal battery and enables the battery to be recharged more than 400 times. The newly developed lithium metal battery boasts 50% higher performance in driving range compared to the existing 600 km.
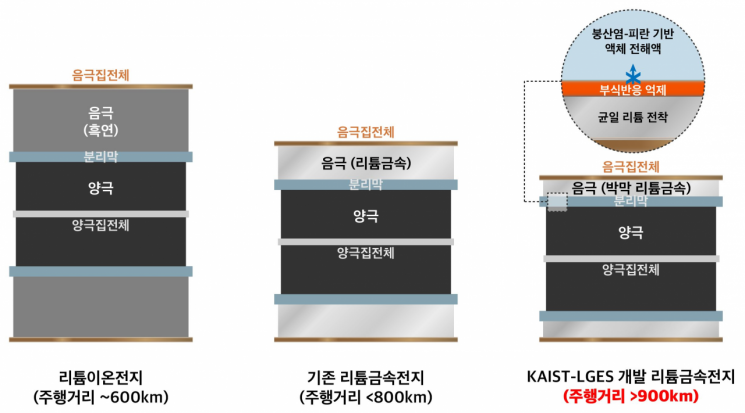
To realize the lithium metal battery, the joint research team applied a 'borate-pyran based liquid electrolyte,' which had not been previously reported, for the first time in the world. Through this, they solved the technical challenges of the lithium metal anode and elucidated the fundamental principles, according to the joint research team.
The borate-pyran electrolyte reconstructs the solid electrolyte interphase (SEI) layer, which is a few nanometers thick on the surface of the lithium metal anode, into a dense structure that blocks corrosion reactions between the electrolyte and lithium.
Although lithium metal batteries have the characteristic of increasing the driving range of electric vehicles, lithium metal has shown technical limitations in securing battery lifespan and stability due to dendrite formation and continuous corrosion caused by liquid electrolytes.
 Infographic on lithium metal battery technology developed by a joint research team from KAIST and LG Energy Solution. Provided by KAIST
Infographic on lithium metal battery technology developed by a joint research team from KAIST and LG Energy Solution. Provided by KAIST
However, the 'SEI restructuring' technology developed by the joint research team simultaneously solves the dendrite and corrosion problems, improving the charge-discharge efficiency of the lithium metal anode, and significantly reducing the weight of the battery anode material and electrolyte compared to existing ones, thereby increasing the energy density.
In particular, the lithium metal battery realized in the study does not require high temperature and pressure during operation, which is also noteworthy as it allows for a simplified battery system design to increase the driving range of electric vehicles.
Professor Hee-Tak Kim of KAIST's Department of Bio and Chemical Engineering evaluated, "The research results of the joint research team are significant in that they visualized the possibility of realizing liquid electrolyte-based lithium metal batteries, which were previously considered impossible."
Also, Dr. Hyuk-Jin Kwon, the first author of the paper and a doctoral candidate, emphasized the significance of the research by stating, "We demonstrated that the limitations of lithium metal batteries can be overcome through nanoscale control of the lithium metal anode interface."
The joint research team's results were also published online in Nature Energy on the 23rd of last month.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
















