'Riblibant' Combination Therapy 'Mariposa'
mPFS 23.7 Months, 30%↑ Compared to Tagrisso
"Establishing a New First-Line Standard Treatment"
FDA Approval Application Within the Year... Approval Expected in 2025
First Domestic Best-in-Class and Blockbuster Drug Anticipated
Yuhan Corporation's non-small cell lung cancer treatment 'Leclaza (Lazertinib)' is raising expectations as the first blockbuster drug in Korea by proving its potential to grow into the best-in-class drug within its category. This is because the combination therapy clinical trial conducted by global big pharma Johnson & Johnson (J&J), which licensed Leclaza, confirmed results surpassing AstraZeneca (AZ)'s standard treatment 'Tagrisso (Osimertinib)'. J&J plans to apply for approval in the U.S. within this year and aims to obtain approval by 2025.
On the 18th, results from the MARIPOSA clinical trial, a combination study of Leclaza and J&J's targeted antibody therapy 'Rybrevant (Amivantamab)', conducted by J&J Innovative Medicine (formerly Janssen), were disclosed. These were released alongside abstracts of key papers to be presented at the 2023 European Society for Medical Oncology (ESMO 2023), held from the 20th to 24th in Madrid, Spain (local time).
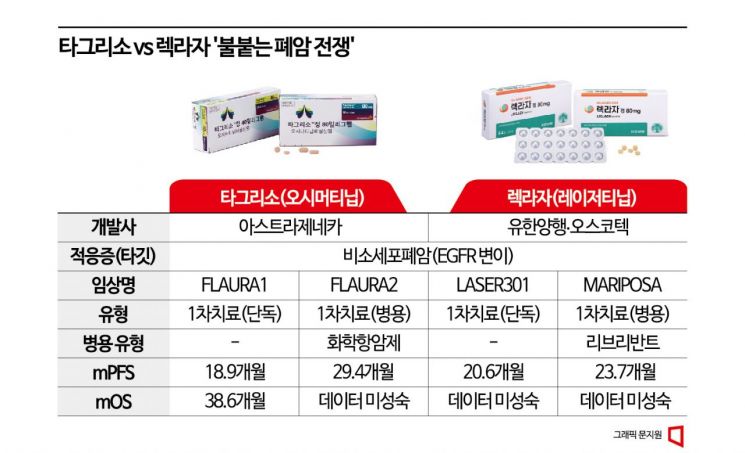
In this trial, the Leclaza-Rybrevant combination group showed a median progression-free survival (mPFS) of 23.7 months, a core indicator for evaluating anticancer drug efficacy, which reduced the risk of disease progression and death by 30% compared to the Tagrisso monotherapy group's 16.6 months. The abstract also confirmed improvements in various detailed indicators and evaluated the MARIPOSA trial as "a clinical study that established the Leclaza-Rybrevant combination therapy as a new standard of care for first-line treatment of epidermal growth factor receptor (EGFR) mutation non-small cell lung cancer."
Currently, the EGFR mutation treatment market has been led by Tagrisso, with the U.S. National Comprehensive Cancer Network (NCCN) recommending it as the preferred therapy. Accordingly, J&J and Yuhan Corporation set the Tagrisso monotherapy group, the existing standard treatment, as the control group in the MARIPOSA trial to compete head-to-head with Tagrisso. Having confirmed results surpassing Tagrisso in this trial, the potential for growth as the best drug in its class has been raised.
If this hope materializes, Leclaza is expected to become Korea's first blockbuster new drug, generating annual sales exceeding $1 billion (approximately 1.353 trillion KRW). J&J's CEO Joaquin Duato named this therapy as one of the key pipelines expected to generate over $5 billion (approximately 6.765 trillion KRW) in annual sales in the future. Domestically, Eugene Investment & Securities analyzed that "considering royalties, the profit generated by Yuhan Corporation from Leclaza could approach 1 trillion KRW by 2030."
 AstraZeneca's non-small cell lung cancer treatment 'Tagrisso (active ingredient Osimertinib)'
AstraZeneca's non-small cell lung cancer treatment 'Tagrisso (active ingredient Osimertinib)' [Photo by Korea AstraZeneca]
However, some analyses suggest that this trial did not produce differentiated results compared to AZ's 'FLAURA2' trial, a Tagrisso and chemotherapy combination therapy prepared to outpace Leclaza. The Tagrisso-chemotherapy combination showed an mPFS of 29.4 months in an independent central review committee (BICR) evaluation, extending mPFS by about 9.5 months compared to the Tagrisso monotherapy group's 19.9 months. Although there were expectations that the MARIPOSA trial's mPFS would surpass this, it did not. One of the key secondary endpoints, objective response rate (ORR), also showed no significant difference, with 86% in the combination group and 85% in the control group.
Nevertheless, detailed indicators suggest that the Leclaza-Rybrevant combination therapy still has sufficient potential to catch up with Tagrisso. Overall survival (OS) analysis is not yet complete, so final results are pending, but interim analysis showed that the Leclaza-Rybrevant combination group demonstrated about 20% improved efficacy compared to the Tagrisso monotherapy group. Since the core of anticancer treatment is to save patients until the end, if a more definitive improvement in OS is proven, the gap could widen again. Additionally, the secondary progression-free survival (PFS) improved by 25%, and the median duration of response (mDoR) was 25.8 months, higher than Tagrisso monotherapy's 16.7 months.
Moreover, due to the high side effects of chemotherapy, the Tagrisso-chemotherapy combination is likely to be preferred by only some patients rather than the entire patient population. J&J also evaluated the MARIPOSA trial during a conference call on the 17th (local time), stating that "it can provide an option free from chemotherapy treatment." J&J plans to apply for FDA approval of the Leclaza-Rybrevant combination therapy within this year.
 2023 European Society for Medical Oncology (ESMO) Annual Congress
2023 European Society for Medical Oncology (ESMO) Annual Congress [Photo by European Society for Medical Oncology]
Additional detailed indicators, including safety results from this trial, will be presented directly by Cho Byung-chul, head of the Lung Cancer Center at Yonsei Cancer Hospital, during the 'Presidential' session, a key presentation at the conference on the 23rd (local time). Of particular interest are the results of the Leclaza monotherapy group. This trial was not only a comparison between the Leclaza-Rybrevant combination group and the Tagrisso monotherapy group but also included a clinical trial of Leclaza monotherapy. Patients were allocated in a 2:2:1 ratio with 429, 429, and 216 patients respectively.
In clinical trials evaluating the efficacy of Tagrisso and Leclaza monotherapies, Tagrisso showed an mPFS of 18.9 months (FLAURA1), while Leclaza showed 20.6 months (LASER 301), indicating Leclaza's superiority. However, LASER 301 used the first-generation 'Iressa (Gefitinib)' as the control group instead of Tagrisso, the third-generation EGFR tyrosine kinase inhibitor (TKI) standard therapy at the time, leading to criticism that it is difficult to confirm superiority over Tagrisso.
Since this trial was designed to confirm the efficacy of the Leclaza-Rybrevant combination therapy, directly comparing the control groups of Tagrisso and Leclaza monotherapies is not scientifically rigorous. Nevertheless, some trends can be observed, and if meaningful results favoring Leclaza monotherapy over Tagrisso monotherapy emerge in this trial, there is potential to prove actual differentiation through subsequent clinical trials.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















