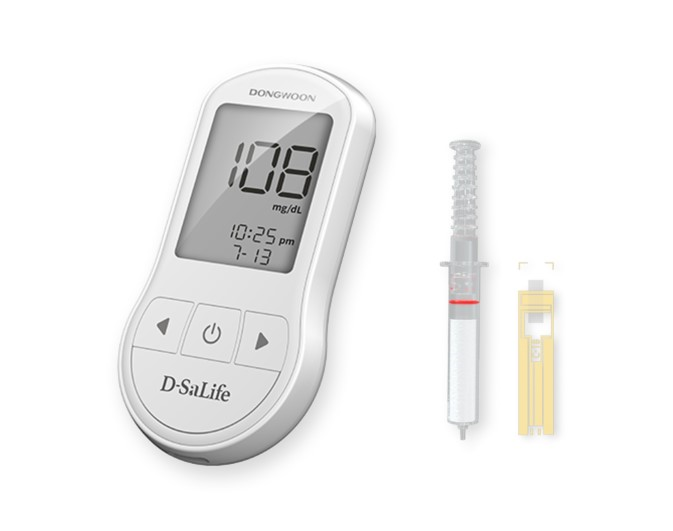
World's First Independently Developed Saliva Glucose Diagnostic Device 'D-SaLife'
Exploratory Clinical Trial on 300 Subjects for 6 Months with Professor Lee Seung-hwan's Team at Seoul St. Mary's Hospital
Professor Lee Seung-hwan: "D-SaLife Accuracy Higher Than Initially Expected"
Dongwoon Anatech announced on the 21st that it achieved successful results in the second exploratory clinical trial of the world's first self-developed saliva glucose diagnostic device, ‘D-SaLife’.
From November last year to April this year, an exploratory clinical trial of D-SaLife was conducted on 300 subjects including healthy individuals, hypoglycemia, prediabetes, and hyperglycemia patients, in collaboration with Professor Seunghwan Lee's research team from the Department of Endocrinology at Catholic University Seoul St. Mary's Hospital.
The top-tier equipment used as the evaluation standard was the hospital-certified blood glucose measuring device ‘YSI 2300 STAT PLUS’, which was used to verify the correlation and validity with D-SaLife. Comprehensive statistical analysis of the test results was conducted by ‘Medical Excellence’, a clinical medical professional service company.
The fasting blood glucose values of 300 subjects measured by D-SaLife showed a Pearson Correlation Coefficient of 92.5% compared to the YSI 2300 standard device measurements, demonstrating high correlation and accuracy.
Among the accuracy evaluation criteria for personal blood glucose measuring devices designated by the International Organization for Standardization (ISO 15197:2013), 90% of patients were included in zone A and 10% in zone B of the Consensus Error Grid, achieving a result where 100% of the 300 blood glucose values fell within zones A and B.
This result is significantly improved compared to the exploratory clinical trial conducted in 2020 with 114 patients by the Department of Endocrinology at Eulji University Hospital in Nowon, Seoul. At that time, 67% of patients were in zone A and the remaining 33% in zone B of the Consensus Error Grid.
Dongwoon Anatech CEO Dongcheol Kim said, "This exploratory clinical trial examined how similar the blood glucose values measured by D-SaLife are compared to the standard device that measures blood glucose through blood," adding, "Since sufficient data meeting the level required by global companies has been secured, we plan to start full-scale overseas license negotiations from the end of this month, aiming to become the world's first saliva glucose diagnostic device."
Professor Seunghwan Lee of Seoul St. Mary's Hospital, who led the exploratory clinical trial, said, "The accuracy of D-SaLife came out higher than initially expected," and added, "Once approved by the Ministry of Food and Drug Safety, it will be a useful device not only for diabetes patients but also for blood glucose management and screening of people in the early stages of diabetes or those with a family history of diabetes."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















