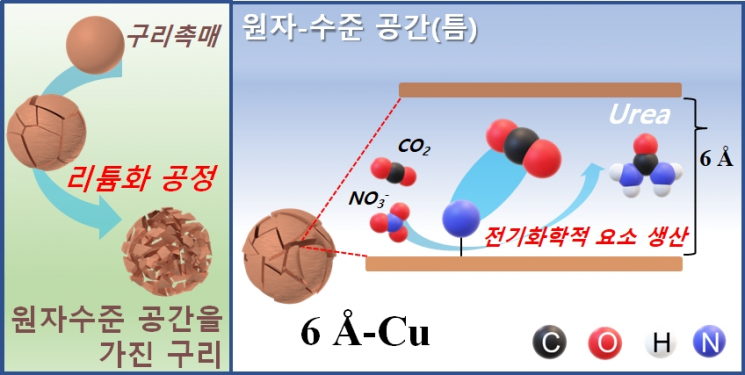
A catalyst capable of reducing greenhouse gas carbon dioxide (CO2) and nitrate (NO3-), a source of fine dust, while producing urea, an essential element for agriculture and various industries, has been developed.
A research team led by Professor Young-Guk Kwon from the Department of Energy and Chemical Engineering at UNIST applied a technology that controls atomic-level gaps within copper to develop a new catalytic material that converts carbon dioxide and nitrate into high value-added urea.
First, Professor Hyun-Gon Song’s team from the Department of Energy and Chemical Engineering implemented atomic-level gaps inside the catalyst through a lithiation process.
Subsequently, Professor Hyun-Wook Lee’s team from the same department observed and identified the formation of atomic-level gaps in real-time using Transmission Electron Microscopy (TEM) analysis.
Through this, they introduced atomic-level gaps between two facets of copper nanoparticles, creating an ideal and efficient catalyst for the electrochemical co-reduction reaction of carbon dioxide and nitrate. They reported a significant improvement in the current efficiency and production rate of urea synthesis using this catalyst.
Among the produced copper catalysts, the copper (6?-Cu) catalyst with a gap distance close to 6 angstroms (?) showed about 17 times faster urea production rate (7541.9 μg h?1 mgcat?1) and more than 19 times higher urea current density (115.25 mA cm?2) compared to conventional copper catalysts.
These figures represent the highest values reported in the academic field to date. Additionally, the catalyst passed a 50-hour stability test, confirming its durability.
The research team revealed through Density Functional Theory (DFT) calculations that the rapid reaction rate of the developed catalyst was due to electron density transfer from the upper to the lower surface of the copper.
They also found that the catalyst stabilized the transition state of the C-N bond, a compound of carbon dioxide and nitrogen, and originated from a low thermodynamic barrier.
Professor Young-Guk Kwon of the Department of Energy and Chemical Engineering stated, “Since the field of electrochemical simultaneous reduction of carbon dioxide and nitrogen compounds is still in its early research stage, expanding fundamental understanding and conducting research on catalytic systems are essential for the commercialization of the technology. We will develop core technology to produce urea domestically, which currently depends on imports, contributing to carbon neutrality.”
Seok-Min Shin, the first author and integrated MS-PhD course researcher, said, “Existing copper catalysts had low selectivity for urea, requiring the development of catalysts that suppress competing reactions and exhibit high selectivity. We developed a high-performance and highly durable urea production catalyst by introducing atomic-level gaps into copper catalysts.”
This study was published online on March 28, 2023, in ‘Energy & Environmental Science,’ a world-renowned journal in the energy and environmental science field, and was selected as an Inside Back Cover paper, published on May 17.
The research was supported by the Mid-career Research Program of the National Research Foundation of Korea under the Ministry of Science and ICT, the Energy Technology Development Project of the Ministry of Trade, Industry and Energy, and the Creative Convergence Research Project of the National Research Council of Science & Technology.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.