KAIST Research Team Uses Naphthalene Diimide Instead of Expensive Vanadium
The current trend in the battery industry is lithium-ion batteries. However, they have chronic issues such as rising raw material costs, resource depletion, fire hazards, and short lifespans. As an alternative to overcome these problems, various batteries made from relatively inexpensive, long-lasting, and stable materials are being developed. Among them, redox flow batteries are considered one of the promising candidates. They feature low cost, low fire risk, and a long lifespan of over 20 years, making them suitable for use as energy storage systems (ESS) linked with renewable energy. However, the recent sharp rise in the price of vanadium, a key material, has become an obstacle to commercialization. A domestic research team has developed a technology to replace vanadium with a cheaper organic molecule.
KAIST announced on the 23rd that a research team led by Professors Byun Hye-ryeong and Baek Moo-hyun from the Department of Chemistry, in collaboration with Professor Seo Jong-cheol’s team at POSTECH, developed a highly soluble and stable organic active molecule for use in aqueous redox flow batteries. Redox is a portmanteau of 'reduction' and 'oxidation.' Redox flow batteries store and release electricity through the 'oxidation-reduction' (reduction-oxidation) reactions of active materials in the electrolyte. They are electrochemical storage devices that store electrical energy as chemical energy in the electrolyte.
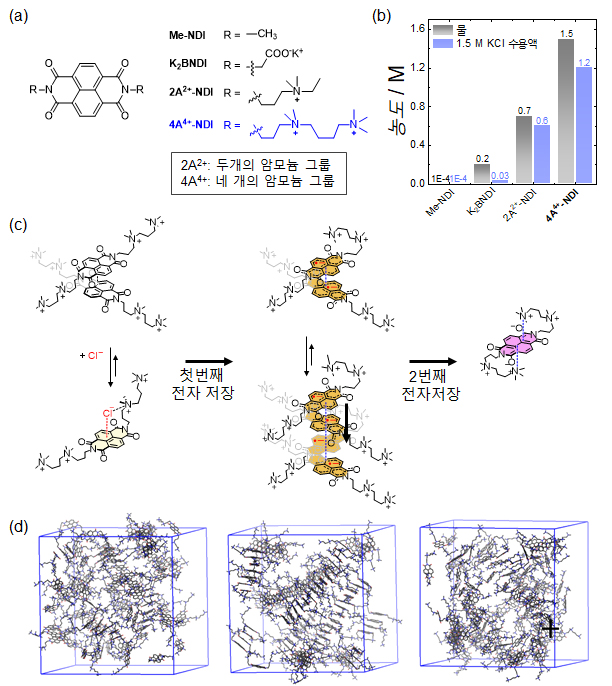 An illustration explaining an organic molecular redox flow battery using naphthalene diimide (NDI) as the active material.
An illustration explaining an organic molecular redox flow battery using naphthalene diimide (NDI) as the active material. (a) Structures of various NDI molecules. (b) Solubility of NDI molecules in water (black bars) and in aqueous electrolyte containing KCl (blue bars). (c-d) Structural changes of the developed NDI molecules upon storing two electrons during two-electron storage. (c) Illustration of cluster bonding and separation of the developed NDI molecules during redox reactions, and (d) MD simulation snapshots. From left to right: prepared NDI molecules, formation of dimer and tetramer clusters after the first reduction reaction, and monomeric stereostructure after the second reduction reaction.
Image source: Provided by KAIST
The research team conducted a study to replace the active materials in redox flow batteries, which are currently dominated by vanadium, with other organic substances. Organic molecules are promising candidates for active materials because their solubility and electrochemical redox potential can be adjusted through various synthetic designs, enabling higher energy storage than vanadium. However, most organic redox active molecules suffer from low solubility or poor chemical stability during redox reactions. Low solubility of active molecules results in low energy storage capacity, and low chemical stability leads to decreased cycle performance. The team used naphthalene diimide (NDI) as the active molecule. Although NDI has high electrochemical stability, it has low solubility in aqueous electrolytes, which has limited research so far.
NDI molecules are almost insoluble in water, but the team introduced four ammonium functional groups to NDI, increasing its solubility up to 1.5 M (1 M = 6.022 x 10^23 active molecules per liter of solution). They also confirmed that when 1 M of the developed NDI molecule was used in a neutral aqueous redox flow battery, about 98% of the capacity was maintained over 500 cycles. This means that only about 0.004% of the capacity decreases per cycle, and after 45 days of operation, only 2% of the initial capacity is lost. Additionally, the developed NDI can store two electrons per molecule, proving that approximately 2 M of electron storage is possible when using 1 M NDI. For reference, the solubility of vanadium, the active material in vanadium redox flow batteries that use high-concentration sulfuric acid solutions, is about 1.6 M, and since each vanadium atom stores one electron, a total of 1.6 M electron storage is possible. Therefore, the developed NDI active molecule can achieve a higher capacity than conventional vanadium.
The research results were published online on the 7th of last month in the international journal Advanced Materials.
Professor Byun Hye-ryeong explained, "We implemented a molecular design principle that allows organic active molecules with low solubility to be used as active materials in redox flow batteries," adding, "This study demonstrated that the chemical reactivity of molecules formed as radicals can be suppressed by utilizing intermolecular bonding forces where molecules bind or separate during redox reactions." She further stated, "In the future, when used in aqueous redox flow batteries, it will be possible to use neutral aqueous electrolytes along with the advantages of high energy density and high solubility," and added, "It is expected to solve corrosion problems caused by the use of acidic solutions in conventional vanadium redox flow batteries."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















