Pfizer Acquires Seegene for $43 Billion
Secures Industry's Top Hot Item 'ADC' Technology
Sanofi Acquires Prevention for $2.9 Billion
FDA Approves First Type 1 Diabetes Delay Drug 'Tigzeld'
Amid warnings about biotech investments following the Silicon Valley Bank (SVB) crisis in the United States, a rare piece of good news emerged that could reverse the mood. On the 13th (local time), global big pharma companies completed two major mergers and acquisitions (M&A) worth a total of $46 billion (approximately 60 trillion KRW) in a single day, reigniting the spark.
On that day, global big pharma companies such as Pfizer and Sanofi announced the acquisitions of cancer drug developer Seagen and diabetes treatment developer Prevention Bio, respectively. Both acquired companies share the commonality of possessing significant technological capabilities, either having recently received FDA approval for new drugs or holding pipelines that have recently attracted industry attention.
Pfizer acquired Seagen at $229 per share, with a total acquisition amount of $43 billion (approximately 56 trillion KRW). Previously, Seagen had engaged in acquisition talks with Merck (MSD) in the U.S. last year, but the deal fell through, leading to Pfizer’s acquisition. This represented a 32.7% premium over the previous day’s closing price.
Seagen currently holds four FDA-approved drugs: three antibody-drug conjugates (ADCs) ? Adcetris, Padcev, and Tivdak ? and Tukysa. The company recorded $1.8 billion in sales last year and is expected to achieve $2.2 billion this year, marking a 12% revenue growth. Pfizer anticipates that Seagen will generate over $10 billion in sales by 2030. Notably, its flagship product Adcetris is also approved and reimbursed in South Korea, and it recorded blockbuster sales of $839 million (approximately 1.095 trillion KRW) last year.
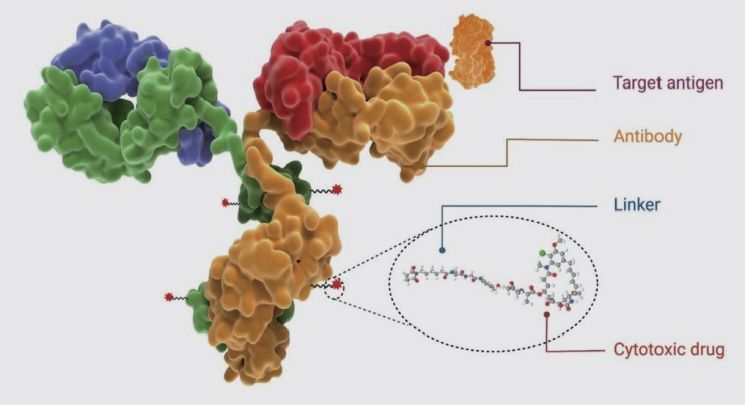
Pfizer’s acquisition of Seagen is analyzed as a move to actively adopt the ADC technology, which has recently gained particular attention. As the name suggests, ADCs are drugs that conjugate antibodies and drugs. By linking an antibody that binds to cancer antigens with a cytotoxic payload drug via a linker, ADCs can deliver toxins specifically to cancer cells like a missile, making them a next-generation cancer treatment technology. Last year, Carolyn Bertozzi, a professor at Stanford University, was awarded the Nobel Prize in Chemistry largely for her work related to ADCs. She was recognized for inventing “click chemistry,” which, when applied to organisms, is called “bioorthogonal chemistry.” This chemistry allows different functional groups to interact only with their designated partners without interacting with other proteins or organic molecules. ADCs were developed based on this principle.
Through this acquisition, Pfizer plans to strengthen its portfolio in the cancer drug field. Chris Boshoff, Pfizer’s Chief Development Officer (CDO) for Oncology and Rare Diseases, stated, “By adding Seagen’s world-class ADC technology, Pfizer will be at the forefront of innovative cancer treatment,” adding, “It will strongly complement our existing portfolio for both solid tumors and hematologic cancers.”
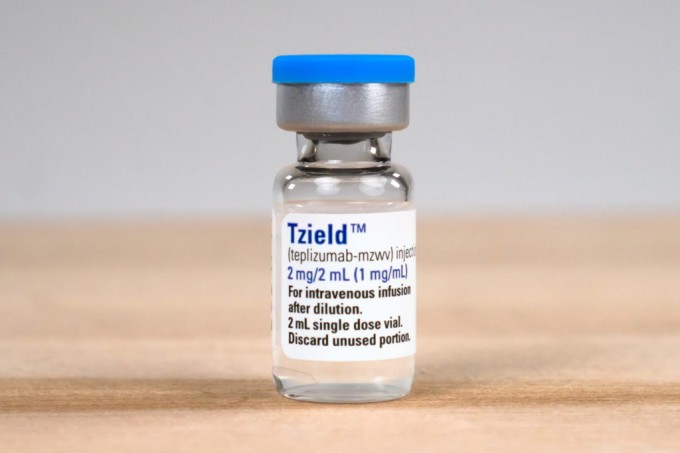 'TG-LED (active ingredient: Teplizumab)', approved by the U.S. Food and Drug Administration (FDA) as a delay agent for the onset of type 1 diabetes
'TG-LED (active ingredient: Teplizumab)', approved by the U.S. Food and Drug Administration (FDA) as a delay agent for the onset of type 1 diabetes [Photo by Prevention Bio]
Prevention Bio, acquired by Sanofi, received FDA approval in November last year for Tzield (active ingredient teplizumab), which delays the onset of type 1 diabetes. It targets CD3 antibodies to reduce T cells associated with autoimmune diseases while increasing regulatory T cells that suppress autoimmune diseases, thereby inhibiting the progression from stage 2 to stage 3 type 1 diabetes.
Unlike type 2 diabetes, which arises from lifestyle issues, type 1 diabetes is a refractory disease in which immune cells attack and destroy pancreatic beta (β) cells, preventing insulin production. Until now, no treatment has been developed. Because insulin secretion is absolutely insufficient, patients must test their blood sugar levels more than four times a day and regularly inject insulin for life. Tzield is known to delay the onset by about two years in clinical trials targeting high-risk groups for type 1 diabetes, with an average of 24.4 months in the control group versus 48.4 months in the treatment group. The longest delay reported in clinical patients is 11 years.
This was the first regulatory approval for a type 1 diabetes onset delay agent. Prior to approval, Sanofi showed expectations by acquiring U.S. rights for Tzield and has now gone further by acquiring the entire company. The acquisition price was $25 per share, totaling $2.9 billion (approximately 3.7851 trillion KRW), representing a 265% premium over the previous day’s closing price.
Especially since this big deal came amid growing concerns about biotechs following the SVB crisis, the market reaction was encouraging. On that day, the stock prices of the acquired companies surged to the acquisition price levels, and major biotech ETFs listed on the U.S. stock market closed up about 3%. Seungmin Kim, a researcher at Mirae Asset Securities, forecasted, “The biotech M&A sentiment improvement will carry more weight than concerns about the SVB crisis.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















