 Purchase procedure for self-treatment cannabis component medicines. [Provided by the Ministry of Food and Drug Safety]
Purchase procedure for self-treatment cannabis component medicines. [Provided by the Ministry of Food and Drug Safety]
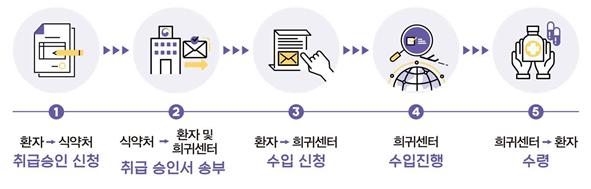
[Asia Economy Reporter Lee Gwan-joo] On the 26th, the Ministry of Food and Drug Safety (MFDS) announced the 'Procedure for Purchasing Cannabis-Derived Medicines for Self-Treatment' to allow the use of cannabis-derived medicines for patients with rare and intractable epilepsy.
To use medicines approved by overseas regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) as cannabis-derived medicines for self-treatment, one must first apply for handling approval from the MFDS and then apply for importation at the Korea Rare and Essential Drugs Center.
Typically, it takes up to 40 days when patients or their guardians apply, but the MFDS is ensuring that cannabis-derived medicines can be received within 10 days to guarantee prompt treatment opportunities for patients.
For the handling application, applicants must submit an application form, a medical diagnosis, medical records, and a medical opinion stating that there are no alternative domestic treatment options to the MFDS. The diagnosis must be prepared by a specialist and include the disease name, medicine name, single dosage amount, daily dosage frequency, total number of dosing days, and method of administration. When applying for handling approval for additional purchases, submission of medical records and opinions is not required. For the import application, the Korea Rare and Essential Drugs Center requires submission of a medicine purchase consent form, transfer contract, and personal information consent form.
Oh Yu-kyung, Commissioner of the MFDS, stated, "We will continue to promote safety innovations to expand and guarantee treatment opportunities for patients with rare and intractable diseases."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















