Sam-il and Samsung Epics Launch Similar 'Ameliboo'
Chong Kun Dang Plans to Release 'Lucenbies' Within the Year
Aging as a Major Cause... Rapid Increase in Patients Due to Aging Population
Original Developer Responds with Mechanism and Convenience Improvements
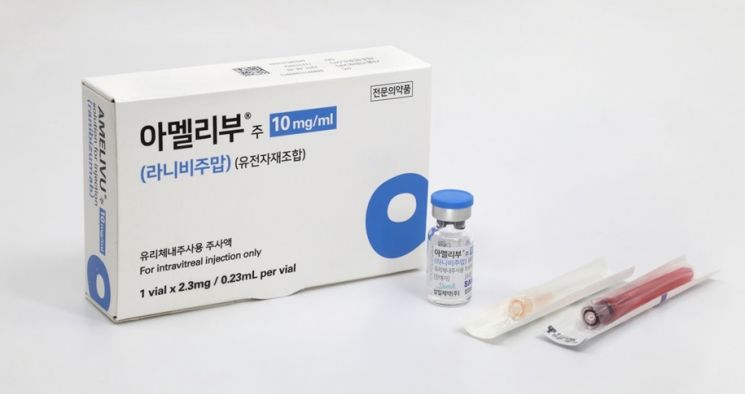 Samsung Bioepis's biosimilar for macular degeneration treatment, 'Amelibu'
Samsung Bioepis's biosimilar for macular degeneration treatment, 'Amelibu' [Photo provided by Samil Pharmaceutical]
[Asia Economy Reporter Lee Chun-hee] The market for macular degeneration treatments, which has seen a recent increase in patients due to age-related diseases, is undergoing rapid upheaval. Since there is no fundamental cure and patients have had to receive periodic eye injections to slow disease progression, the burden on patients has been significant. Amid this, biosimilars (biopharmaceutical generics) with significantly reduced prices have entered the market, prompting original developers to engage in additional research and development to maintain their market share.
According to industry sources on the 26th, Samil Pharmaceutical recently began full-scale sales of 'Amelivuz', a biosimilar of 'Lucentis' (active ingredient ranibizumab), a treatment for macular degeneration and diabetic macular edema developed by Samsung Bioepis. This follows a domestic distribution and sales agreement signed between the two companies in June last year.
Amelivuz currently holds full-label approval in South Korea for all indications recognized under the National Health Insurance for Lucentis, including ▲neovascular (wet) age-related macular degeneration ▲diabetic macular edema ▲retinal vein occlusion macular edema ▲choroidal neovascularization. Starting this year, the Ministry of Health and Welfare has decided to recognize insurance coverage for ranibizumab biosimilars other than Lucentis, so there is expected to be little difficulty in entering the market. Furthermore, CKD Pharmaceutical's biosimilar 'Lucenbes (CKD-701)' also received approval for all related indications last year and is expected to be launched soon, intensifying competition among biosimilars.
Macular degeneration is a disease where waste products accumulate in the macula, the central part of the retina, initially causing dry macular degeneration. This progresses to wet macular degeneration due to excessive blood vessel growth, potentially leading to blindness. Also known as age-related macular degeneration or 'senile macular degeneration,' its prevalence sharply increases with aging and is considered one of the top three causes of blindness among the elderly. With global aging trends, the number of patients is expected to continue rising. Data analysis firm GlobalData has estimated that the market size for wet macular degeneration treatments in nine major countries including the U.S., Germany, Japan, China, and Australia will reach $18.7 billion (approximately 23 trillion KRW) by 2028.
Treatment for macular degeneration involves directly injecting antibody drugs such as Lucentis or Regeneron's 'Eylea' (active ingredient aflibercept) into the eye. These drugs bind to vascular endothelial growth factor (VEGF)-A to inhibit neovascularization and slow disease progression. Although Lucentis was launched first, Eylea has rapidly captured the market by offering dosing convenience, requiring injections every two months instead of monthly like Lucentis.
However, since both drugs require periodic injections, the costs are substantial. Even with health insurance coverage, off-label use of 'Avastin' is reportedly common in South Korea. Avastin, originally an anticancer drug, has no approved indication for macular degeneration, but patients often bear a lower out-of-pocket cost with Avastin than with the insured Lucentis or Eylea, whose copayments are higher or similar after insurance coverage.
The introduction of biosimilars is expected to significantly reduce this burden. Typically, biosimilars are priced 30-40% lower than the original drugs. In fact, Amelivuz's insurance price is 463,773 KRW, 43% lower than Lucentis's 820,636 KRW. Samsung Bioepis is also rapidly targeting the U.S. market with a similar strategy, pricing their biosimilar 40% lower than the original.
In response, original developers are continuing research to maintain their market share by improving mechanisms of action and enhancing dosing convenience to reduce patients' time and cost burdens. Roche, which owns Lucentis through its subsidiary Genentech, received approval from the Korean Ministry of Food and Drug Safety on the 20th for 'Vabysmo' (active ingredient faricimab), a treatment for neovascular macular degeneration. Unlike Lucentis or Eylea, which inhibit VEGF-A, Vabysmo is a humanized bispecific antibody drug that also blocks the angiopoietin (Ang)-2 pathway to inhibit neovascularization. After the initial four doses administered every four weeks, if disease activity is absent, dosing is extended to once every 16 weeks, significantly reducing treatment burden. Clinical trials confirmed non-inferior visual improvement compared to Eylea.
Regeneron is also focusing on follow-up developments to maintain its market share. Amid competition with biosimilars and Vabysmo, Eylea's sales in Q4 last year fell to $1.5 billion (approximately 1.8548 trillion KRW), marking the first year-over-year decline and raising urgent concerns. Regeneron is currently conducting clinical trials to increase Eylea's dose from 2 mg to 8 mg, aiming to extend dosing intervals from 8 weeks to 16 weeks. Clinical data confirmed this higher dose is not inferior to the current Eylea treatment.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















