Binding Two Different Antigens Simultaneously
Enhancing Therapeutic Effect While Reducing Costs
Six Approved Bispecific Antibodies... Growing Unmet Needs
 Lee Jae-sun, Executive Director of Samsung Biologics (right), and Choi Hyung-seok, Head of Research Planning Team at the Bio Research Institute, are introducing Samsung Biologics' bispecific antibody platform 'S-DUAL' at the 'Bio Knowledge Concert' held online on the 6th.
Lee Jae-sun, Executive Director of Samsung Biologics (right), and Choi Hyung-seok, Head of Research Planning Team at the Bio Research Institute, are introducing Samsung Biologics' bispecific antibody platform 'S-DUAL' at the 'Bio Knowledge Concert' held online on the 6th. [Photo by Samsung Biologics]
[Asia Economy Reporter Lee Gwan-joo] The global competition to develop ‘bispecific antibody’ new drugs is intensifying. Not only global big pharma but also domestic bio companies have entered the field, with over 600 pipelines reportedly under development worldwide. The market potential is large, and since bispecific antibodies can overcome the limitations of existing single-antibody-based therapies, the pharmaceutical and bio industries are expected to continue their challenges.
Maximizing Therapeutic Efficacy and Increasing Patient Convenience
Typical antibodies act on only one target antigen. In contrast, bispecific antibodies can bind to two different antigens simultaneously. The concept of bispecific antibodies was first proposed 50 years ago, but due to manufacturing issues and other challenges, it faced failures until the rapid advancement of biotechnology led to the first approval relatively recently in 2015.
The biggest feature of bispecific antibodies is their therapeutic efficacy. A single drug targets two antigens and can maximize effects through a sophisticated mechanism of action design. They effectively prevent drug resistance through dual targeting, improve targeting ability, and reduce off-target toxicity. Another feature is cost reduction. Compared to using two or more drugs, patient convenience can be increased, and development costs are also reduced.
So far, a total of six bispecific antibodies have been approved worldwide. Four have been approved by the U.S. Food and Drug Administration (FDA), and two by the European Medicines Agency (EMA). They are mainly being developed as immuno-oncology drugs for non-small cell lung cancer, lymphoma, multiple myeloma, etc., but this year, Genentech’s ‘Vabysmo’ in the U.S. was approved as the first ophthalmic bispecific antibody (for macular degeneration), expanding indications.
Fierce Domestic and International Development Competition
As bispecific antibodies gain attention, global competition is heating up. The large unmet needs and the relatively few approved products make market entry highly attractive to many companies. According to Roots Analysis, a pharmaceutical market research firm, the global bispecific antibody drug market is expected to reach $9.3 billion (approximately 12.9 trillion KRW) by 2030. Among global big pharma, Roche, Pfizer, and AbbVie are active, while domestically, ABL Bio, AbClon, PharmAbcine, Y-Biologics, and Celltrion are accelerating their efforts.
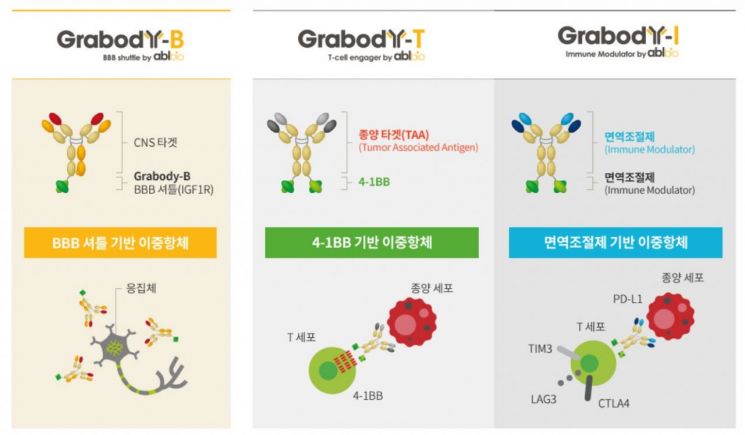 ABL Bio's three self-developed bispecific antibody platforms. 'Grabody-B' (left) is a bispecific antibody platform that enhances blood-brain barrier (BBB) penetration and is utilized in the development of bispecific antibody therapeutics for degenerative brain diseases. 'Grabody-T' is a platform that selectively binds to the CRD4 region, maximizing 4-1BB tumor-specific activation. Lastly, 'Grabody-I' is an immunomodulator-based bispecific antibody used in the development of solid tumor therapeutic candidates.
ABL Bio's three self-developed bispecific antibody platforms. 'Grabody-B' (left) is a bispecific antibody platform that enhances blood-brain barrier (BBB) penetration and is utilized in the development of bispecific antibody therapeutics for degenerative brain diseases. 'Grabody-T' is a platform that selectively binds to the CRD4 region, maximizing 4-1BB tumor-specific activation. Lastly, 'Grabody-I' is an immunomodulator-based bispecific antibody used in the development of solid tumor therapeutic candidates.ABL Bio owns three bispecific antibody platforms independently. Based on these, the bispecific antibody candidate ‘ABL301’ for degenerative brain disease treatment recently signed a joint development and technology transfer agreement with Sanofi, and ‘ABL111’ was designated as an orphan drug for gastric cancer treatment by the FDA, showing promising results.
Celltrion signed a joint development contract with U.S.-based Abpro for the bispecific antibody therapy ‘ABP102’ targeting HER2-positive breast cancer. Celltrion secured global joint development and sales rights for ABP102 and will be responsible for animal efficacy studies, process development, preclinical animal studies, clinical development, and commercialization of the candidate.
Contract Development and Manufacturing Organizations (CDMOs) are also responding swiftly, seeing bispecific antibodies as a new market. Samsung Biologics launched the next-generation bispecific antibody platform ‘S-DUAL.’ This platform innovatively improves the weaknesses of existing bispecific antibody platforms. The Y-shaped bispecific antibody’s two arms are configured asymmetrically to ensure stability and enable rapid detection of issues. It enhances the binding strength of bispecific antibodies, reduces impurity rates, and achieves a high purity of up to 99%.
Choi Hyung-seok, head of research planning at Samsung Biologics’ Bio Research Center, emphasized, “Antibody experts have evaluated the asymmetric structure as a ‘smart idea,’” and added, “We are confident that we have sufficient global competitiveness.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















