National Institute of Health Stem Cell Bank
Announces Checklist for Reducing Cell Therapy Production Costs
[Asia Economy Reporter Chunhee Lee] The National Stem Cell Bank of the Korea Disease Control and Prevention Agency's National Institute of Health announced on the 30th that it has published a checklist for ‘reducing production costs’ of cell therapies in collaboration with international advanced biopharmaceutical experts.
Cell therapies are treatments developed using living cells, tissues, and the like. They are expected to be therapeutic options for diseases that are difficult to treat with conventional medicines, such as rare and intractable diseases.
Business intelligence firm Visiongain projected that the global advanced regenerative medicine market, encompassing cell therapies, gene therapies, and tissue engineering therapies, will rapidly grow from $27.3 billion (approximately 39 trillion KRW) in 2018 at an average annual growth rate of 22%, expanding to $298 billion (approximately 427 trillion KRW) by 2030. In particular, the Asian market is expected to grow more than 20 times during the same period, from $3.3 billion (approximately 4.7275 trillion KRW) to $72.2 billion (approximately 103 trillion KRW).
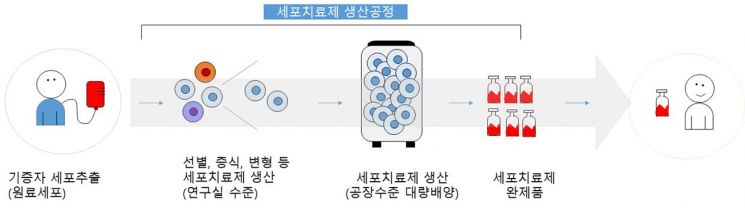
Although cell therapy technology advances rapidly into clinical stages, production experience is still limited, causing many companies and researchers handling cell therapies to experience trial and error, which often leads to increased production costs. Accordingly, the National Institute of Health, based on production experience of ‘starting material’ cells used in cell therapies and therapeutic production, jointly with international experts, published ‘items to consider for cost reduction’ with case examples.
This announcement summarizes the contents of the ‘International Stem Cell Banking Initiatives’ workshop held in Osong, Chungbuk, hosted by the National Institute of Health in 2019. Detailed information was published on the 29th in the internationally renowned academic journal Nature’s sister journal, npj Regenerative Medicine.
Dr. Ohad Karnieli of Avda Biotechnology in Israel, an international advanced biopharmaceutical expert, stated, “Chimeric antigen receptor (CAR)-T cells currently incur very high production costs due to their nature as personalized autologous therapies. However, if some production processes are automated, cells for multiple patients can be produced simultaneously, reducing costs.” Professor Yuji Arakawa of Kyoto University in Japan also explained, “Producing one batch (about 300 vials) of GMP-grade induced pluripotent stem cell (iPSC) lines costs approximately 300 million KRW,” adding, “Producing cell therapies by obtaining GMP-grade iPSC lines from an iPSC bank can reduce costs.”
Other scholars suggested strict quality control regarding cell origin and source verification, contamination and cell transformation, thorough validation of research results, establishment of cell banks, systematic documentation, and comparison of advantages and disadvantages between in-house production and external contract manufacturing to reduce costs.
Dr. Junwook Kwon, Director of the National Institute of Health, said, “We expect that these expert opinions will contribute to promoting the practical application of advanced biopharmaceutical development, which is a national agenda.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















