Expert: "Removal of 'Jwijot' Not Possible with Cosmetics or Quasi-Drugs"
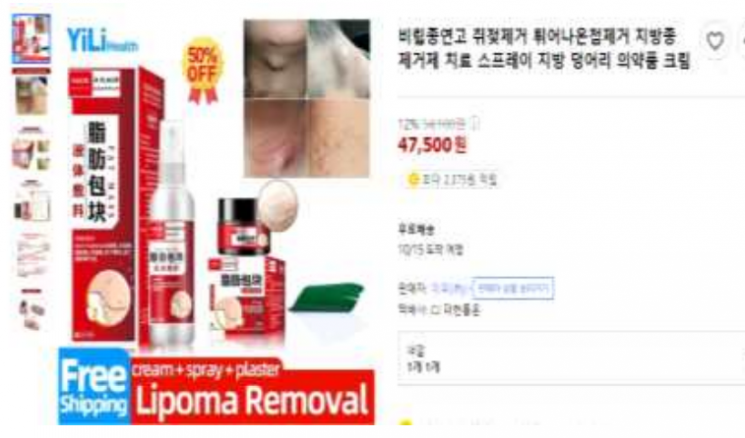 The Ministry of Food and Drug Safety (MFDS) has identified 569 cases of advertising and selling online with claims of removing skin tags. Photo by MFDS
The Ministry of Food and Drug Safety (MFDS) has identified 569 cases of advertising and selling online with claims of removing skin tags. Photo by MFDS
[Asia Economy Reporter Yoon Seul-gi] The Ministry of Food and Drug Safety (MFDS) has detected 569 cases of online advertisements and sales claiming to remove skin tags after conducting a focused inspection.
According to the MFDS on the 28th, there are currently no pharmaceuticals, cosmetics, medical devices, or quasi-drugs approved in Korea for the removal of skin tags. Therefore, advertising and selling products claiming to remove skin tags is illegal, and the online distribution and sale of pharmaceuticals is also illegal. Skin tags are benign tumors that occur on the skin due to keratinocyte and collagen proliferation and do not significantly affect health.
Of the 569 cases detected, 300 involved illegal online sales and advertisements of unapproved foreign pharmaceuticals, 148 were cosmetic advertisements claiming medical efficacy and effects using images of skin tag removal ointments or milia removal, and 115 involved sales and advertisements of general products that could be mistaken for medical devices with skin tag removal efficacy.
The MFDS consulted a private advertising verification group composed of experts who verify unfair advertisements of food and drugs regarding these product advertisements. The experts emphasized that skin tags cannot be removed with cosmetics or quasi-drugs that have minimal impact on the human body, and that the efficacy and effects of devices that remove skin tags by blocking blood supply have not been verified.
The MFDS urged consumers, saying, "Using unverified products to remove skin tags by yourself may cause complications and side effects such as contact dermatitis and skin infections," and advised, "You should consult and receive treatment from a dermatologist."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
















