Expectations for the DTx Era to Open Within the Year
Commercialization Difficult Without Health Insurance Coverage
Challenges Such as Patient Participation and Acceptance by Medical Staff and Institutions Must Be Overcome
 On the 1st, attendees are engaged in a discussion at the 'Digital Therapeutics (DTx) Overseas Expansion Strategy Seminar' held at The-K Hotel Seoul in Seocho-gu, Seoul. Photo by Lee Chunhee spring@
On the 1st, attendees are engaged in a discussion at the 'Digital Therapeutics (DTx) Overseas Expansion Strategy Seminar' held at The-K Hotel Seoul in Seocho-gu, Seoul. Photo by Lee Chunhee spring@
[Asia Economy Reporter Lee Chun-hee] It is expected that the first domestically produced digital therapeutic (DTx) will be released as early as this year. While anticipation is growing for the full-fledged opening of the DTx era, experts point out that thorough preparation is necessary, as various challenges still need to be overcome in the process of insurance reimbursement and commercialization even after passing the hurdle of approval from the Ministry of Food and Drug Safety (MFDS).
According to the industry on the 2nd, at the ‘Digital Healthcare Global Capacity Building Seminar’ held yesterday at The-K Hotel Seoul in Seocho-gu, Seoul, hosted by the Korea Health Industry Development Institute and the Korea Smart Healthcare Association, diverse opinions were exchanged regarding DTx approval, reimbursement, and overseas expansion.
Health Insurance Reimbursement Is the Biggest Challenge
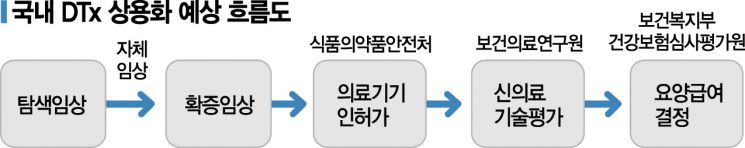
Among the DTx currently under development, a total of 13 have received clinical trial plan approval from the MFDS. Among these, companies such as Welt, NewNaps, LifeSemantics, AimMed, and Hi are in the final clinical phase known as the ‘confirmatory clinical trial’ stage, raising expectations that the first DTx could be launched within this year. While the industry commonly refers to them as ‘therapeutics,’ the MFDS defines DTx as ‘software as a medical device (SaMD) that provides evidence-based therapeutic interventions to patients to prevent, manage, or treat medical disabilities or diseases.’ Unlike therapeutics, which are understood as pharmaceuticals, DTx is classified as a medical device and even termed ‘digital therapeutic device.’
Kim Young, CEO of Synex, emphasized the need to "go back to basics," stressing that to overcome the MFDS approval hurdle, attention must be paid to the fact that DTx is a medical device. Kim said, "Many DTx developers come from software backgrounds rather than the medical industry, so they feel overwhelmed," but suggested a solution: "They must properly establish whether it is a medical device, its classification within medical devices, and clinical trials, and continuously consult with the MFDS."
Even after obtaining MFDS approval, many hurdles remain. Since DTx is also a product, revenue generation through sales is necessary. However, in Korea, where a strong public health insurance system called National Health Insurance is established, health insurance is virtually a single purchaser. Unlike countries such as the United States, where private insurance dominates and business-to-business (B2B) sales for workplace insurance are possible, commercialization is difficult without passing the reimbursement hurdle.
The Health Insurance Review & Assessment Service (HIRA) is also preparing to present guidelines on the application of health insurance to DTx within this year. Shim Bo-ram, a deputy research fellow at HIRA conducting related research, explained, "From the perspective of health insurance, policies must be implemented for the entire population with limited resources," adding, "While the technical value is fully recognized, the focus is on whether it is worthy of reimbursement and how to evaluate its value."
Currently, the decision on reimbursement for DTx is likely to require passing the ‘New Health Technology Assessment’ conducted by the National Evidence-based Healthcare Collaborating Agency (NECA). Since it is a novel technology, another procedure to verify safety and efficacy must be undertaken. Shim said, "We are considering proving clinical efficacy through the innovative medical technology track," and added, "We are reviewing a plan to allow early entry with a preliminary listing at about a 90% patient co-payment rate, followed by stricter evaluation during formal listing."
Higher Acceptance Needed for Therapeutic Effectiveness
Participants pointed out that after reimbursement, the challenge of the ‘real world’ must also be overcome. Most DTx currently under development are in the form of cognitive behavioral therapy (CBT). Since treatment is conducted through the participation of various patients, the success of treatment depends on whether patients can participate at a high level over a long period. In fact, for ‘Reset-O’ by Pear Therapeutics, the first DTx approved by the U.S. Food and Drug Administration (FDA), results showed that treatment effectiveness varied from 48% to 89% depending on how much of the program was completed.
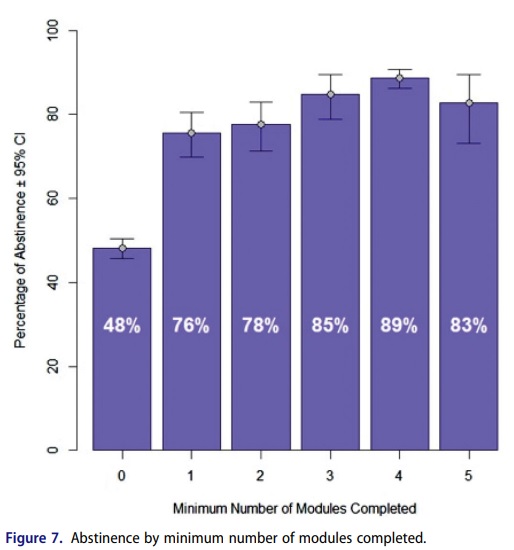 Therapeutic effects of Pear Therapeutics' 'Reset-O' by the number of module completions. / Image source=CMRO Journal
Therapeutic effects of Pear Therapeutics' 'Reset-O' by the number of module completions. / Image source=CMRO Journal
Shim said, "Since DTx is used by patients after prescription, treatment effectiveness can vary depending on participation," and added, "We are also considering the need to evaluate usability when deciding on reimbursement." Min Jung-sang, CEO of Emotive, which is developing a DTx for attention deficit hyperactivity disorder (ADHD), said, "We developed a game-type DTx as a result of considering usability," adding, "Since DTx requires long-term interaction, we chose a method that can be accessed without disrupting the daily lives of children with ADHD."
There was also an opinion that, as a prescribed medical device, medical staff and institutions need to consider their acceptance of prescribing DTx. Jeon Hong-jin, professor of psychiatry at Samsung Medical Center, said, "DTx cannot be prescribed just because a doctor wants to; it requires a coding agreement between the hospital and the company," and pointed out, "If hospitals and doctors do not receive income such as fees and only the DTx developer gains revenue, there is no incentive to prescribe." Shim added, "We are considering basic consultation fees and education counseling fees generated by DTx as medical service fees," and "We are also looking into developing additional fees."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















