Selection of Regulatory Improvement Tasks in Food and Medical Fields
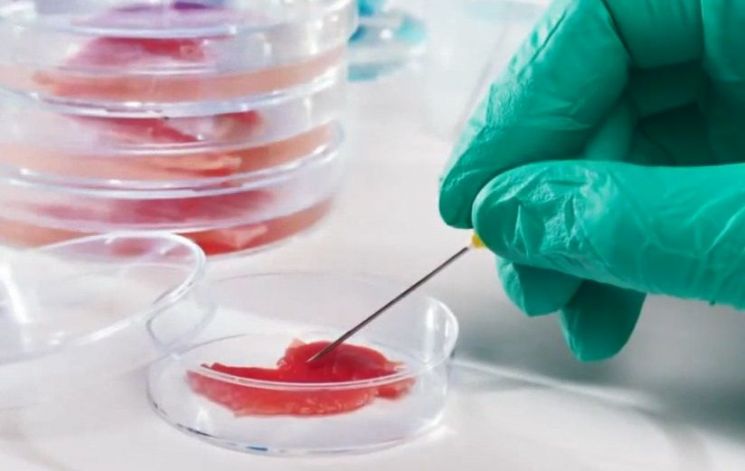
[Asia Economy Reporter Oh Hyung-gil] Cultured meat produced through cell culture is expected to be recognized as a food ingredient.
The Korea Chamber of Commerce and Industry (KCCI) and the Ministry of Food and Drug Safety (MFDS) selected and announced the "Top 100 Regulatory Innovation Tasks in the Food and Pharmaceutical Sectors" on the 11th to support the growth of new industries in the food and pharmaceutical fields and to resolve corporate difficulties caused by regulations.
The regulatory innovation tasks announced that day include 19 tasks supporting new industries, 45 tasks improving public inconvenience and burdens, 13 tasks for international harmonization, and 23 tasks resolving procedural regulations, totaling 100 tasks in the food sector and the medical device and pharmaceutical sectors. By sector, there are 50 tasks in the food sector and 50 in the pharmaceutical sector.
The KCCI stated, "Both organizations agreed that unnecessary regulations must be boldly improved to enable the rapid market launch of innovative products and to strengthen the global competitiveness of the food and pharmaceutical industries," adding, "The regulatory innovation tasks announced this time are significant in that they were jointly discovered and selected by the government and the business community through active communication, which will enhance the sense of regulatory innovation felt at the corporate level."
They decided to expand the "temporary recognition of food ingredients" to allow food ingredients applying new technologies to be recognized. Through this regulatory improvement, foods using cell culture technology will be included in the recognition target, which is expected to help companies entering the future food industry.
In particular, growth in the cultured meat business, which produces meat by harvesting and culturing animal muscle cells without pain, is anticipated. According to global consulting firm A.T. Kearney, the global meat market is expected to grow to a scale of 2,000 trillion KRW by 2040, with cultured meat accounting for 35% of that market.
 Korea Chamber of Commerce and Industry, Ministry of Food and Drug Safety, and Korea Consumer Agency jointly held the "100 Major Tasks for Regulatory Innovation in Food and Drug" briefing on the 11th at the Korea Chamber of Commerce and Industry Hall on Sejong-daero, where Kang Seok-gu, Head of the Investigation Headquarters at the Korea Chamber of Commerce and Industry, is giving the briefing.
Korea Chamber of Commerce and Industry, Ministry of Food and Drug Safety, and Korea Consumer Agency jointly held the "100 Major Tasks for Regulatory Innovation in Food and Drug" briefing on the 11th at the Korea Chamber of Commerce and Industry Hall on Sejong-daero, where Kang Seok-gu, Head of the Investigation Headquarters at the Korea Chamber of Commerce and Industry, is giving the briefing.
The "Smart Label (QR Code) Utilization for Food Information Display" allows food information that must be displayed on product containers or packaging under current law to be replaced with a QR code. This greatly improves readability by prominently displaying essential information and replaces the rest with a QR code, reducing packaging replacement costs. Additionally, by linking with the government-operated "Food Information Platform," changes in food information can be notified without cost.
Pharmaceutical regulatory improvements include restructuring medical device classification and testing standards and transitioning the natural and organic cosmetics certification system to private sector leadership.
The "Customized Rapid Classification System for Medical Devices" was selected as a task. Previously, when launching newly developed medical devices, there was no "item classification," so approval applications had to be submitted using "similar classification" after consultation with the MFDS, complicating administrative procedures. However, by comparing risk, intended use, and performance of similar products, devices can be classified as "temporary items" for rapid approval. Low-risk software medical devices, such as those analyzing blood pressure and electrocardiogram data, will be exempt from MFDS clinical trial plan approval, simplifying the clinical trial approval process.
The government-led natural and organic cosmetics certification system will be transitioned to private sector leadership. Unlike the previous system where certification was conducted by MFDS-designated institutions, various private certification standards can now be utilized, reducing certification costs and promoting more active product launches.
Kang Seok-gu, head of the KCCI headquarters, said, "Active regulatory relaxation efforts by the government are necessary, and the KCCI will continue to identify difficulties experienced by companies on the ground and work closely with the MFDS to propose improvements to the government."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.