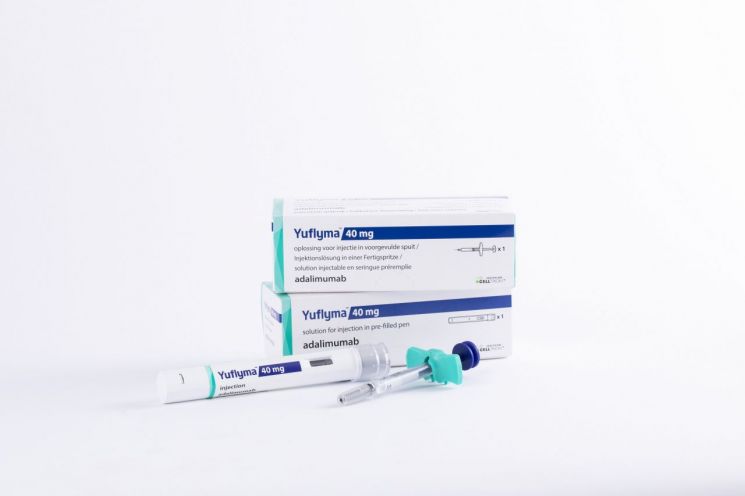
Celltrion Applies for Clinical Trial on 'Yuflyma' as a Substitute for 'Humira'
Strategy to Expand Market Share by Proving Equivalence
Active Development of Superior 'Biobetters'
[Asia Economy Reporter Lee Chun-hee] As competition in the biosimilar (biopharmaceutical generic) market intensifies, movements surrounding the development of ‘interchangeable biosimilars’ are becoming more active. In addition, strategies aiming for market dominance through ‘biobetters’ that outperform the original products are also being pursued.
Celltrion Applies for Phase 3 Clinical Trial of Interchangeable Biosimilar
According to industry sources on the 3rd, Celltrion recently submitted a global Phase 3 clinical trial plan to the U.S. Food and Drug Administration (FDA) to secure interchangeability for ‘Yuflyma (CT-P17)’, a biosimilar of ‘Humira’. The trial will compare pharmacokinetics, efficacy, and safety between multiple crossover dosing groups of Yuflyma and Humira and a Humira maintenance dosing group in 366 patients with plaque psoriasis. Through this, Celltrion plans to secure the status of interchangeable biosimilar for Humira and expand its market share.
The importance of interchangeable biosimilars lies in the characteristics of biosimilars. While generic drugs, which are copies of synthetic drugs, must prove biological equivalence, biosimilars must prove similarity. Due to the nature of biopharmaceuticals, it is practically impossible to make them exactly identical to the original product because the cells used in manufacturing differ, and the manufacturing processes such as culture conditions and purification methods also vary. For example, Celltrion’s ‘Inflectra (the U.S. brand name of Remsima)’, a biosimilar of ‘Remicade’ (active ingredient infliximab), and Samsung Bioepis’s ‘Renflexis’ are both labeled by the FDA with distinct ingredient names ‘infliximab-dyyb’ and ‘infliximab-abda’, respectively.
However, if approved as an interchangeable biosimilar, it must prove equivalence rather than similarity, which facilitates smoother market entry. The FDA designates a biosimilar as interchangeable if it can demonstrate that the biosimilar and the original product produce the same clinical result. The label explicitly states interchangeability, allowing pharmacists to substitute the interchangeable biosimilar for the original without the prescribing physician’s intervention. Moreover, even if the original developer tries to maintain market share by expanding indications, the interchangeable biosimilar can obtain approval for other indications of the original through ‘extrapolation’ based on equivalence, enabling easier response.
"Surpass the Original" Challenge
However, among the 36 biosimilars approved by the FDA so far, only two?Siltyzeo (a Humira biosimilar) and Semglee (a Lantus biosimilar)?have been recognized as interchangeable biosimilars, indicating that gaining this recognition is not easy.
This is why Celltrion is pursuing the designation of Yuflyma as an interchangeable biosimilar of Humira. Humira is a mega blockbuster drug consistently ranking among the top global sales. Last year, it recorded sales of $20.694 billion (approximately 27 trillion KRW). It held the world’s top sales position for nine consecutive years until 2020. Although Pfizer’s COVID-19 vaccine ‘Comirnaty’ took the throne last year with $36.8 billion (approximately 48 trillion KRW) in sales, Humira is likely to regain the top spot next year. Besides Siltyzeo, seven biosimilars including Amjevita (Amgen), Hyrimoz (Sandoz), Hadlima (Samsung Bioepis), Abrilada (Pfizer), Hulio (Mylan), and Yusimry (Coherus) have been commercialized based on FDA approval, but they still maintain strong sales in the U.S. market due to patent protections and other factors.
However, since Celltrion has already reached an agreement with AbbVie to allow U.S. sales of Yuflyma starting July next year, if Yuflyma obtains FDA approval expected within this year and subsequently gains interchangeable biosimilar designation, it is expected to secure a favorable position for market entry.
Not only securing equivalence but also actively developing biobetters that improve upon original drugs is underway. Biobetters refer to drugs that improve administration methods, frequency, side effects, or demonstrate superior efficacy compared to the original. Celltrion’s ‘Remsima SC’ is a representative example. Celltrion first biosimilarized the autoimmune disease treatment Remicade as an intravenous (IV) form ‘Remsima IV’ and then developed a biobetter in the form of a subcutaneous injection (SC). It is the only SC formulation product including the original and biosimilars, and SC is generally known to offer better dosing convenience and safety than IV, emerging as a new treatment option.
GC Green Cross is also actively engaged in continuous research and development (R&D), having improved the efficacy of the Hunter syndrome treatment ‘Hunterase’ compared to existing treatments and recognized it as a biobetter, as well as developing an additional intracerebroventricular (ICV) formulation that can be directly injected into the brain ventricle.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















