Korea Investment & Securities Report
[Asia Economy Reporter Minji Lee] On the 13th, Korea Investment & Securities forecasted that SCM Lifescience, a developer of stem cell therapeutics, will demonstrate its technological capabilities through upcoming clinical results.
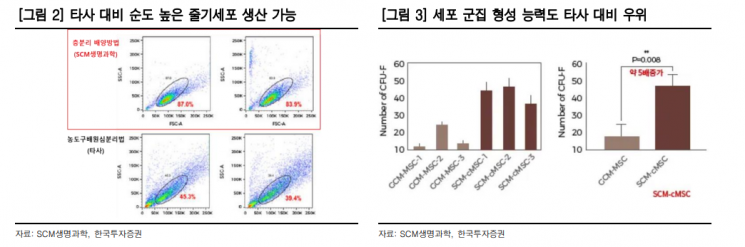
SCM Lifescience is a developer of stem cell therapeutics. Using its proprietary stratified culture method, it isolates and cultures bone marrow-derived mesenchymal stem cells (MSCs) to build a high-purity stem cell library. Since cell culture proceeds without artificial manipulation under serum-free media conditions, the cluster formation ability of the isolated stem cells is superior compared to other companies.
They have a process capable of producing up to approximately 3,000 doses through a 3D bioreactor. This production method is about 10 times improved compared to the conventional 2D technique, enabling cost reduction and supply matching demand after clinical success. To actively prepare for clinical development and commercialization, they plan to construct cGMP-level production facilities.
The clinical results will be a noteworthy factor in maintaining the technological superiority of the stratified culture method. Clinical efficacy results are important because MSCs have no safety issues. The first notable clinical trial is a Phase 2 study targeting patients with atopic dermatitis. It is scheduled to conclude by the end of this year, with clinical results to be announced in the first quarter of next year. Dupixent, prescribed for severe atopic dermatitis patients, forms a global market worth 9 trillion KRW this year. The downside is that the annual drug cost per patient reaches about $30,000, and it must be administered once every two weeks.
Researcher Jaeju Wi from Korea Investment & Securities said, “SCM Lifescience’s pipeline has cases where efficacy lasted up to two years after three doses at two-week intervals, showing drug price competitiveness,” adding, “The domestic rights for this indication were licensed to Handok, and Phase 3 clinical trials and commercialization will be conducted by Handok.”
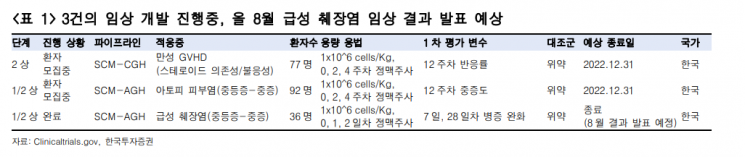
The second clinical trial to watch is the Phase 2a study for acute pancreatitis patients, with results announced this August. Designated as an orphan drug in the development stage, conditional product approval application is possible within 2025 depending on Phase 2b results.
The company’s entry into the immuno-cell therapy market is also positive for both its performance and stock price. Researcher Wi explained, “They secured the CAR-CIK pipeline from CoImmune, which showed a 78% complete remission rate in acute lymphoblastic leukemia and met the primary endpoint of safety,” adding, “They also plan to expand indications to solid tumors through a dendritic cell-based pipeline, so it is necessary to pay attention to related momentum.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















