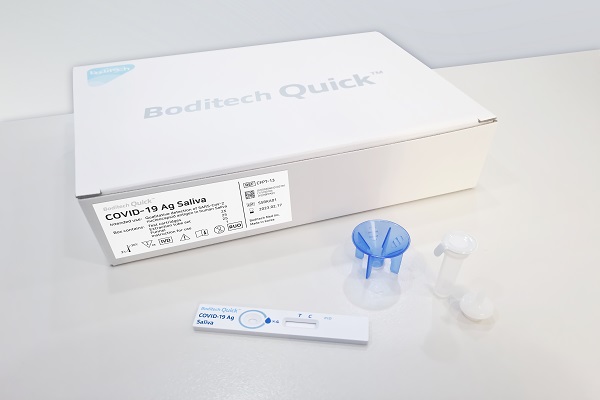
[Asia Economy Reporter Lee Gwanju] Boditech Med announced on the 3rd that it has obtained overseas export approval from the Ministry of Food and Drug Safety for its COVID-19 saliva self-test kit.
The newly approved 'Boditech Quick COVID-19 Ag Saliva' met the MFDS standards with a sensitivity of 90% and specificity of 100%. The company explained that it reduced the cost by more than half compared to existing products, adding price competitiveness to the convenience of the product.
The saliva self-test kit involves placing saliva into a tube containing buffer solution, mixing it, and then applying it to the test kit. Instead of inserting a swab into the nose, the specimen is collected using saliva, eliminating pain and enhancing testing convenience.
Choi Eui-yeol, CEO of Boditech Med, said, “The kit shows superior specificity and sensitivity compared to existing tests for major current variants such as Omicron and Stealth Omicron, and its improved testing convenience has attracted significant interest overseas. Since we have obtained overseas export approval, we expect domestic use approval to proceed quickly once MFDS requirements are met.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.