MFDS Approves PCL's Saliva-Based COVID-19 Self-Test Kit
[Asia Economy Reporter Kim Young-won] From now on, COVID-19 testing using saliva instead of nasal swabs has become possible.
The Ministry of Food and Drug Safety announced on the 29th that it has approved the first COVID-19 antigen self-test kit in Korea that uses saliva as the specimen.
The newly approved product is PCL's 'PCL SELF TEST - COVID19 Ag.'
This product met the self-test kit approval standards with a sensitivity of over 90% and specificity of over 99%. Unlike the existing nine self-test kits that collect nasal (nasal cavity) specimens, it uses saliva from inside the mouth as the specimen.
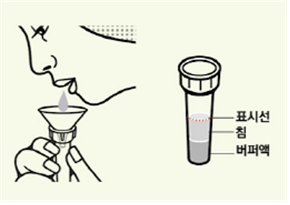
The saliva kit consists of a paper funnel, a solution container, and a filter cap. It is used by spitting saliva into the solution container using the funnel and mixing it with the extraction solution.
An official from the Ministry of Food and Drug Safety stated, "The approval of the saliva self-test kit is expected to help improve ease of use for children, the elderly, and others."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
















