The Pandemic Nears Its End... "Find New Opportunities"
[Asia Economy Reporter Lee Gwan-joo] Domestic pharmaceutical and bio companies that grew during the COVID-19 pandemic are accelerating efforts to find future growth paths. They are actively engaging in research and development (R&D) and investment to discover new revenue sources. They are also speeding up their entry into overseas markets.
SK Bioscience, Full-Scale Preparation for the ‘Next Pandemic’
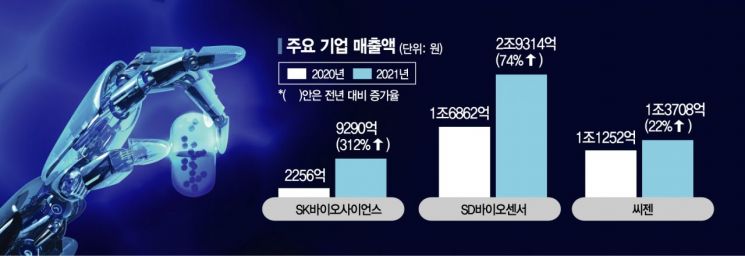
According to the pharmaceutical and bio industry on the 22nd, domestic companies that gained attention with vaccines and diagnostic kits during the COVID-19 phase are accelerating the establishment of management strategies to prepare for the post-COVID era. A representative example is SK Bioscience. Following the production of AstraZeneca (AZ) vaccine, Korea’s first COVID-19 vaccine, they are currently producing the Novavax vaccine (Nuvaxovid), and have continued their growth by gaining global attention through the development of ‘GBP510,’ which is expected to be the first domestically produced COVID-19 vaccine.
SK Bioscience is leveraging the global network it has built in this process to activate its ‘Next Pandemic’ response strategy. First, it plans to expand its business areas through contract development and manufacturing organization (CDMO) services in new fields such as cell and gene therapy (CGT), and launch a ‘Glocalization’ project to establish production infrastructure in various countries. It will also enhance the performance of its key products, including Skycellflu, the world’s first quadrivalent cell-cultured influenza vaccine; Skyzoster, the world’s second shingles vaccine; and Skyvaricella, a chickenpox vaccine that recently won a bid in the world’s largest procurement market, PAHO. Additionally, it plans to acquire new vaccines.
They have also established strategies aligned with the endemic phase of COVID-19. Alongside GBP510, they plan to continuously secure competitiveness by developing multivalent vaccines that can respond to various variants, combo vaccines targeting both influenza and COVID-19 simultaneously, and universal vaccines targeting sarbecoviruses. In particular, they have embarked on developing an innovative drug in the form of a ‘nasal spray’ that prevents viral infection by being sprayed into the nose. This project is supported by research and development funding from the Bill & Melinda Gates Foundation and involves collaboration with overseas research institutions such as the International AIDS Vaccine Initiative (IAVI) and the Institute for Protein Design (IPD) at the University of Washington.
SD Biosensor and Seegene Expand Overseas Markets
SD Biosensor, which has transformed into a global in vitro diagnostics specialist, is considered one of the biggest beneficiaries of COVID-19. Riding the surge in demand for COVID-19 diagnostic kits, SD Biosensor recorded sales of 2.9314 trillion KRW and operating profit of 1.3698 trillion KRW last year, becoming the healthcare company with the highest sales in Korea. Based on this, SD Biosensor is seeking future growth paths through active mergers and acquisitions (M&A) and investments. At the end of last month, it acquired the German in vitro diagnostics distributor ‘Bestbion’ for 16.1 billion KRW in its first M&A deal of the year. Bestbion has established a distribution network capable of 24-hour delivery across Germany and Austria and distributes hundreds of immunodiagnostic, microbiological, and molecular diagnostic products. SD Biosensor believes this approach allows more effective and time-saving entry into the European market compared to establishing local subsidiaries.
SD Biosensor also plans to invest 188 billion KRW to expand production facilities and equipment at its Chungbuk Jeungpyeong plant. Through this, it aims to newly produce 57 million units annually of the rapid molecular diagnostic device ‘STANDARD M10’ cartridges at the Jeungpyeong plant by next year. The STANDARD M10 is a key product that SD Biosensor regards as a future growth engine; it is a rapid molecular diagnostic device that maintains the accuracy of polymerase chain reaction (PCR) testing while reducing the testing time to under one hour. They plan to launch diagnostic products for various diseases including COVID-19, human immunodeficiency virus (HIV), hepatitis C virus (HCV), and multidrug-resistant tuberculosis (MDR-TB).
Molecular diagnostics specialist Seegene is also pursuing large-scale R&D and expansion into global markets. It is particularly focusing on entering the U.S. market. In February, it recruited molecular diagnostics expert Richard Krieger as CEO of its U.S. subsidiary, followed by the hiring of Dr. Glenn Hanson, a bio-clinical expert to oversee the medical science division, intensifying product development aimed at the U.S. market. Recently, it has accelerated new market development by obtaining certifications in Australia and Europe for ‘Allplex™ RV Master Assay,’ a product capable of simultaneously testing 19 respiratory viruses including COVID-19, influenza, and adenovirus. An industry insider commented, “It is a natural step for companies that grew during the COVID-19 situation and have investment capacity to leverage their strengths to pioneer new markets,” adding, “Government and industry-level strategies to respond to the post-COVID era will also be established soon.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.


![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















