Vaccination for Children Aged 6 Months to Under 5 Years
Expected to Begin as Early as March
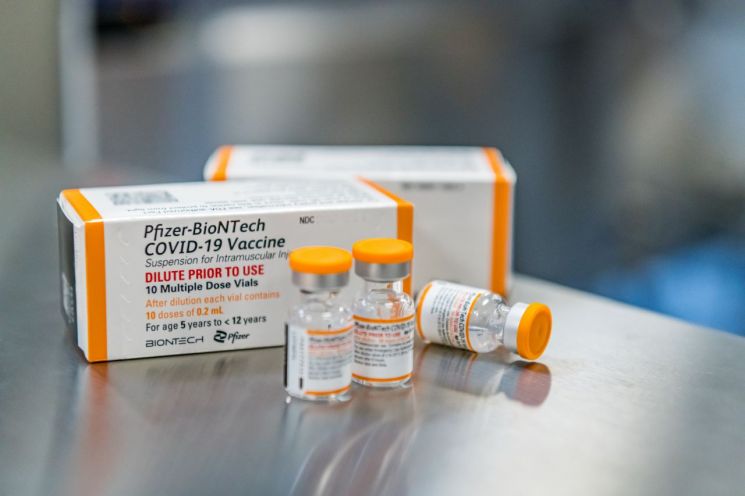
[Asia Economy International Department Reporter] On the 1st (local time), U.S. pharmaceutical company Pfizer and German BioNTech requested the U.S. Food and Drug Administration (FDA) to expand the emergency use authorization of the COVID-19 vaccine to children under 5 years old.
According to CNBC and other broadcasts, the emergency use authorization application submitted on this day targets children from 6 months to under 5 years old. Currently, only children aged 5 and older can receive the vaccine in the U.S. Sources familiar with the matter said this application for children under 5 was unusually expedited at the FDA's request, ahead of the original schedule.
Pfizer and BioNTech have begun submitting the data required for emergency use authorization to the FDA, and both companies stated that this process is expected to be completed within a few days.
The FDA is reviewing this application through a fast-track procedure, and according to AP, vaccinations for children aged 6 months to 4 years could begin as early as March.
The FDA plans to hold an advisory committee meeting composed of external experts in mid-month regarding Pfizer's application.
Albert Bourla, CEO of Pfizer, mentioned the recent spread of the Omicron variant in a statement, saying, "As hospitalizations of children under 5 due to COVID-19 surge, our mutual goal with the FDA is to prepare for future waves of other variants and provide parents with options to help protect their children."
The plan is to administer only one-tenth of the adult vaccine dose to children under 5 years old.
Once final approval is granted by the FDA and other health authorities, these children will initially receive two doses of the COVID-19 vaccine, but depending on the final clinical trial results, a third dose is likely to be administered.
In low-dose vaccine clinical trials, Pfizer and BioNTech found that children aged 2 to 4 did not generate sufficient immune responses as expected, so they are conducting studies on a third dose for this age group.
If the safety and efficacy of the third dose are confirmed in additional clinical trials, Pfizer and BioNTech plan to apply to the FDA for emergency use authorization of a third vaccine dose for children under 5 years old.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















