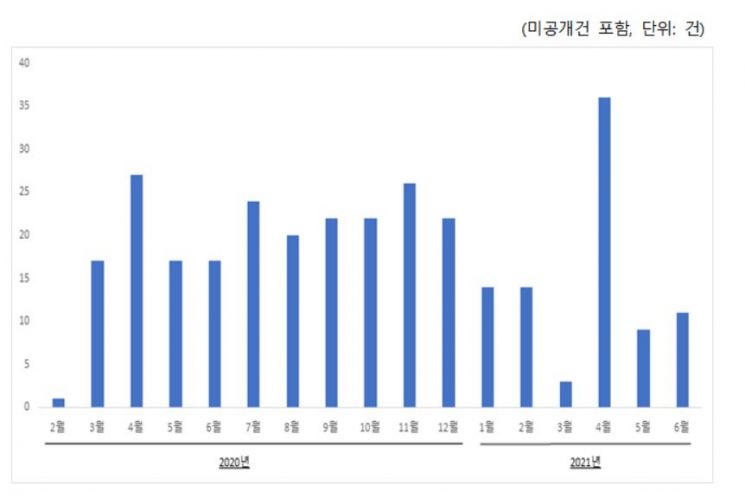
 Graph of monthly patent application trends for COVID-19 treatments from February 2020 to June 2021. Provided by the Korean Intellectual Property Office.
Graph of monthly patent application trends for COVID-19 treatments from February 2020 to June 2021. Provided by the Korean Intellectual Property Office.
[Asia Economy (Daejeon) Reporter Jeong Il-woong] As the development competition for COVID-19 treatments intensifies among global pharmaceutical companies, with treatments being seen as game changers, related patent applications are also active alongside treatment development.
◆ Domestic and international pharmaceutical companies focus on treatment development and clinical trials = According to the Korean Intellectual Property Office on the 12th, recently global pharmaceutical companies are eagerly participating in clinical trials for the development of COVID-19 treatments.
Recently, the U.S. FDA granted emergency use authorization for 11 treatments including intravenous drugs from Eli Lilly, Regeneron, Genentech, GSK, and Gilead’s Remdesivir. Among these, Remdesivir received formal approval for use.
U.S. Merck (MSD) is currently focusing on developing the oral treatment Molnupiravir, and it is expected to receive FDA emergency use authorization within this year depending on clinical trial results.
Additionally, Roche, known for developing Tamiflu, and Pfizer, known for vaccine development, are also conducting clinical trials for oral COVID-19 treatments.
In South Korea, Celltrion received conditional approval for the country’s first COVID-19 treatment, ‘Rekkironaju,’ in February, and domestic treatment development centered on pharmaceutical companies and research institutes is in full swing.
◆ Competitive treatment development leads to active patent applications = As competition for COVID-19 treatment development intensifies, related patent applications have also become active. Pharmaceutical companies and research institutes engaged in treatment development are seeking to secure rights through patents.
In South Korea, from February last year to June this year, a total of 302 patents related to COVID-19 treatments were filed, of which 13 have been registered for their antiviral effects against COVID-19.
This includes one patent approved as a COVID-19 treatment (Rekkironabu), two patents currently undergoing clinical trials (such as Dongwha Pharmaceutical’s new drug derived from Buglossoides arvensis), and one patent with completed clinical trials (Bukwang Pharmaceutical’s Levovir).
Examining COVID-19 treatment patents filed domestically by applicant type, companies such as pharmaceutical firms account for 147 cases (48.7%), the highest number, followed by government agencies and affiliated research institutes with 66 cases (21%), and universities with 55 cases (18%).
Notably, patent applications based on national research and development projects total 78 cases, accounting for 25.8% of the total. This indicates that with government support, government agencies, research institutes, universities, and companies are actively engaged in both treatment development and patent applications.
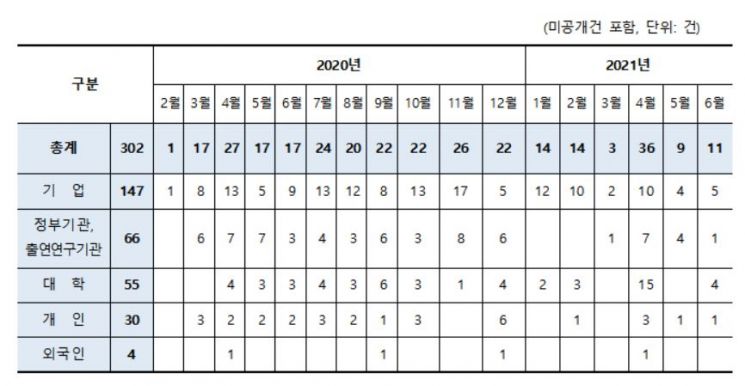 Monthly Patent Application Status by Applicant Type for COVID-19 Therapeutics. Provided by the Korean Intellectual Property Office.
Monthly Patent Application Status by Applicant Type for COVID-19 Therapeutics. Provided by the Korean Intellectual Property Office.
Earlier, the government launched the ‘Pan-Government Support Committee for COVID-19 Treatment and Vaccine Development’ in April last year to support the development of domestic COVID-19 treatments.
Gwak Hee-chan, an examiner at the Pharmaceutical Chemistry Examination Division of the Korean Intellectual Property Office, said, “To end COVID-19, the development of treatments that can be easily taken is urgently needed,” adding, “For this purpose, domestic and international pharmaceutical companies are actively developing COVID-19 treatments through new substance development, antibody treatments, drug repositioning, and other methods.”
He continued, “The increase in related patent applications alongside COVID-19 treatment development is also a clear phenomenon,” but added, “However, even if a drug obtains patent registration, it must undergo safety and efficacy evaluations based on clinical trial results and receive approval from the Ministry of Food and Drug Safety, so careful attention to post-approval procedures is necessary.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















