SK Bio, Genexine, and 3 Other Companies
All-Out Effort to Secure mRNA Vaccine Technology
[Asia Economy Reporter Seo So-jung] The government has decided to support domestic developers' entry into Phase 3 clinical trials in the second half of the year and to pursue advance purchases in order to accelerate the development of domestically produced COVID-19 vaccines.
On the 25th, the government held the 10th meeting of the Pan-Governmental Support Committee for COVID-19 Therapeutics and Vaccine Development to discuss support measures for COVID-19 vaccine development in the second half of this year and the current status and support plans for domestic messenger ribonucleic acid (mRNA) vaccine technology.
The government plans to promote advance purchases by comprehensively considering immunogenicity, safety, likelihood of success, production capacity, and ease of vaccination for companies that have entered Phase 3 clinical trials or have obtained interim results from Phase 2 trials. Domestic companies have long demanded that the government actively engage in advance purchases to establish vaccine sovereignty and promote the development of K-vaccines.
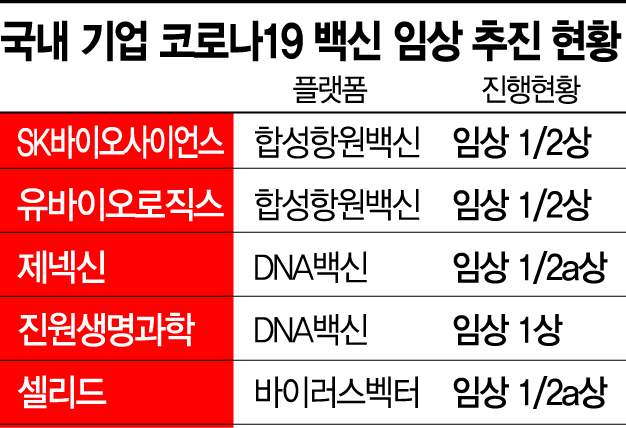
Currently, five domestic vaccine developers?SK Bioscience, YuBioLogics, Genexine, GeneOne Life Science, and Cellid?are aiming to progressively enter Phase 3 clinical trials starting in the second half of the year. SK Bioscience is scheduled to begin Phase 3 trials next month, and Genexine is also securing funds to enter Phase 3 trials.
The government plans to provide full support as the five companies aim to enter Phase 3 clinical trials in the second half of this year. Although the budget for vaccine clinical support was increased from 49 billion KRW last year to 68.7 billion KRW this year, the government will also pursue additional budget allocations if necessary.
The operation of the National Central Institutional Review Board (Central IRB) will also be expedited. Through integrated multi-center reviews, this will resolve the inconvenience of having to obtain approval from each medical institution’s Institutional Review Board (Institutional IRB) and support rapid entry into clinical trials.
Preparations such as specimen analysis infrastructure for Phase 3 clinical trials are being pushed to be completed within the first half of this year. To recruit participants for large-scale clinical trials, the National Clinical Trial Support Foundation is conducting pre-recruitment of vaccine clinical trial participants, and once Phase 3 trials are in full swing, participants will be actively connected.
Domestic mRNA Vaccine Technology Lags Overseas by Three Years... Cross-Ministerial Collaborative Support
Additionally, recognizing that domestic mRNA vaccine technology lags behind overseas by about three years, the government has decided to provide cross-ministerial collaborative support for 39 technology demands across seven fields to close the gap.
As a result of the first and second rounds of technology demand surveys conducted among domestic companies and academia, demands were identified in seven areas: antigen design and optimization, raw material production, IVT vector and mRNA production, vaccine delivery system production such as lipid nanoparticles (LNP), purification, mass production, and efficacy evaluation. Approximately ten companies plan to proceed with clinical trials in the future.
The government is also promoting the ‘Next-Generation Infectious Disease Response Platform Core Technology Development Project.’ This project supports the development of next-generation core technologies covering the entire infectious disease response cycle?from spread prediction to rapid diagnosis, treatment, and prevention?and will support 12 tasks over four years from this year through 2024.
Kang Do-tae, Vice Minister of Health and Welfare, stated, "We will strive to visualize results early through cross-ministerial full support for vaccine Phase 3 clinical trials in the second half of this year," adding, "Through focused cross-ministerial support to secure mRNA vaccine technology, we will advance Korea’s bioindustry and prepare for the post-COVID-19 era."
The domestic bioindustry welcomed the government’s strengthened support but collectively voiced the need for more comprehensive backing ahead of the costly Phase 3 clinical trials. A representative from a domestic vaccine developer said, "Conducting global Phase 3 clinical trials requires thousands of billions of KRW, including operational and material costs, which poses a significant risk for individual companies to bear," adding, "With global vaccination expanding, it is crucial to proceed swiftly into Phase 3 trials, and more active support must be provided."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















