"'Heterologous Vaccination' Related Overseas Studies Show No Safety or Immunity Issues"
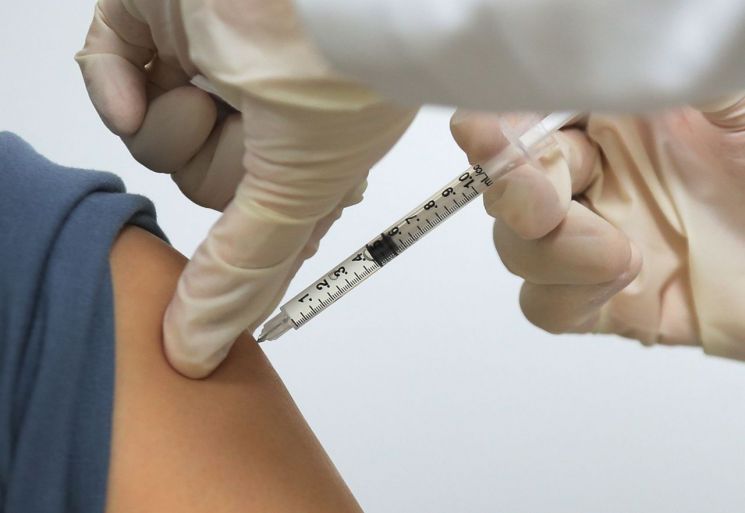
[Asia Economy Reporter Kim Ji-hee] The government has decided to proceed with 'heterologous vaccination' by administering the Pfizer vaccine for the second dose to those scheduled to receive the AstraZeneca (AZ) vaccine in July.
On the 17th, Jeong Eun-kyung, Commissioner of the Korea Disease Control and Prevention Agency (KDCA), stated at the regular COVID-19 briefing, "Originally, about 830,000 doses of the AstraZeneca vaccine imported through COVAX were scheduled to be introduced by the end of June and used for the second dose, but the schedule has been changed to after July." She added, "Accordingly, after deliberation by the Vaccination Expert Committee, it was decided to administer the second dose with the Pfizer vaccine to some recipients."
The Vaccination Expert Committee also decided that while COVID-19 vaccinations should ideally be administered with the same vaccine, based on overseas cases and research results, and considering vaccine supply situations, heterologous vaccination can be conducted if necessary by matching the interval of the first dose vaccine. Commissioner Jeong explained, "Considering supply schedules, distribution, and transportation periods, to proceed with the second dose stably, it was judged that changing to Pfizer vaccination at entrusted medical institutions for about one month in July would allow for more stable management of the situation, leading to the decision for heterologous vaccination." She added, "The expert committee and specialists also agreed that proceeding with heterologous vaccination was more appropriate in terms of effectiveness than delaying the schedule."
According to the authorities, monitoring overseas research results shows that heterologous vaccination has higher immune efficacy and no safety issues. Countries currently allowing heterologous vaccination include Canada, Sweden, Germany, France, Finland, and Italy. A study in Spain found that when AZ-Pfizer heterologous vaccination was administered, binding antibodies increased 30 to 40 times and neutralizing antibodies increased sevenfold compared to a single AZ dose. A UK heterologous vaccination study reported that mild side effects such as fatigue and injection site pain increased, but no serious adverse reactions were observed.
Accordingly, those who received their first dose with the AZ vaccine at entrusted medical institutions from mid-April will temporarily receive their second dose with the Pfizer vaccine in July. However, for those eligible for heterologous vaccination who wish to receive the second dose with the AZ vaccine, separate guidance for AZ vaccination will be provided. The vaccination interval will be maintained at 11 to 12 weeks according to the AZ vaccine schedule, and recipients can receive their vaccination at the scheduled institution and date without changing their reservation. However, for medical institutions that do not administer the Pfizer vaccine, a change of vaccination institution will be necessary, and separate notifications will be sent to the recipients.
The plan for second doses after August will be reviewed comprehensively based on vaccine supply status, domestic and international research results, and overseas cases. Commissioner Jeong stated, "Efforts are underway to advance the delivery schedule of individually contracted AZ vaccine doses earlier than originally planned, and these individually contracted doses are expected to be introduced from mid-July."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















