Passed 1st Review by MFDS One Month After Application
'Triple Screening'... Formal Approval Expected Within This Month
86% Prevention Effect in Elderly, 91% in Patients with Underlying Conditions
Safety "At an Acceptable Level"
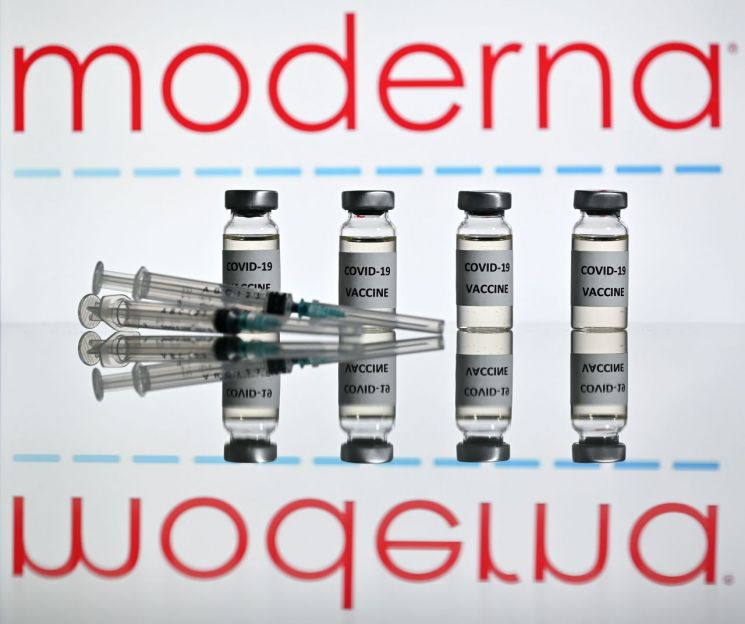 mRNA (messenger ribonucleic acid) COVID-19 vaccine developed by Moderna in the United States
mRNA (messenger ribonucleic acid) COVID-19 vaccine developed by Moderna in the United States [Image source=AFP Yonhap News]
[Asia Economy Reporter Lee Chun-hee] Moderna's COVID-19 vaccine has passed the first verification by the Ministry of Food and Drug Safety (MFDS).
On the 10th, the MFDS announced that at the verification advisory committee meeting held the previous day, based on clinical trial data of the Moderna vaccine, it was judged that the preventive effect for approval is acceptable. This comes about a month after GC Green Cross, responsible for the domestic approval process and distribution of the Moderna vaccine, applied for import product approval on the 12th of last month.
The MFDS reviews through a triple advisory process involving the COVID-19 vaccine and treatment approval verification advisory committee, the Central Pharmaceutical Affairs Deliberation Committee, and the final inspection committee. Seven experts, including infectious disease specialists, vaccine experts, and clinical statistics experts, attended this advisory committee meeting.
If the Moderna vaccine receives final product approval, it will become the fourth vaccine approved domestically following AstraZeneca (AZ), Pfizer, and Janssen vaccines. The Moderna vaccine, like the Pfizer vaccine, was developed using an mRNA (messenger ribonucleic acid) platform that induces an immune response against the COVID-19 virus. It requires two doses and ultra-cold chain distribution at -25 to -15 degrees Celsius is essential. Unlike viral vector vaccines such as AZ and Janssen, which have been associated with rare blood clot controversies, no major side effects have been reported so far. Approximately 20 million doses (40 million shots) are planned to be introduced domestically. However, the specific introduction schedule has not yet been finalized.
94.1% Prevention Rate After 14 Days... Consistent Effectiveness in Elderly and Patients with Underlying Conditions
 Kim Sang-bong, Director of the Bio-Drug Division at the Ministry of Food and Drug Safety, is announcing the results of the verification advisory committee meeting regarding the progress of the product approval for the COVID-19 vaccine developed by Moderna on the morning of the 10th at the MFDS briefing room in Cheongju, Chungbuk.
Kim Sang-bong, Director of the Bio-Drug Division at the Ministry of Food and Drug Safety, is announcing the results of the verification advisory committee meeting regarding the progress of the product approval for the COVID-19 vaccine developed by Moderna on the morning of the 10th at the MFDS briefing room in Cheongju, Chungbuk. [Image source=Yonhap News]
The verification advisory committee announced that based on Phase 3 clinical trial data conducted in the United States, the Moderna vaccine showed high preventive efficacy. The clinical trial involved a total of 30,351 participants, including 6,817 (22.5%) with underlying conditions and 7,520 (24.8%) elderly aged 65 or older.
According to the clinical trial results, 11 out of 14,134 vaccinated individuals were confirmed COVID-19 positive 14 days after the second dose, compared to 185 out of 14,073 in the placebo group, showing a 94.1% preventive effect. The vaccine showed consistent effectiveness regardless of age or underlying conditions, with 86.4% efficacy in those aged 65 and older and 90.9% in patients with underlying conditions.
In immunogenicity assessments, an indirect indicator of vaccine effectiveness, the Moderna vaccine also demonstrated efficacy. The seroconversion rate, which is the proportion of individuals whose antibody titers increased more than fourfold compared to before vaccination, was 100% for neutralizing antibodies that bind to the COVID-19 virus and induce preventive effects.
The advisory committee also judged the vaccine's safety to be at an acceptable level. Most local and systemic reactions after vaccination disappeared within three days. The frequency and severity of reactions in the elderly were lower than in adults.
However, nine cases of 'serious adverse drug reactions' that could not be ruled out as related to the vaccine were reported. These included two cases of facial swelling, and one case each of nausea, vomiting, rheumatoid arthritis, autonomic nervous system imbalance, peripheral edema, dyspnea, and B-cell small lymphocytic lymphoma. Kim Hee-sung, head of the MFDS Rapid Review Division, explained, "The two cases of facial swelling had a history of filler procedures within six months, and the B-cell small lymphocytic lymphoma case was a 75-year-old cancer patient with a history of metastatic lung or breast cancer. Other cases had similar histories, and most were recovering at the time of clinical trial data submission."
The MFDS will receive the second advisory review from the Central Pharmaceutical Affairs Deliberation Committee on the 13th and announce the results that afternoon. Since the MFDS has announced plans to drastically shorten the COVID-19 vaccine approval and review period to within 40 days, a formal approval decision is expected within this month.
 Moderna's job posting for a PV Director at its subsidiary in Korea on its official website (Image source: Moderna homepage)
Moderna's job posting for a PV Director at its subsidiary in Korea on its official website (Image source: Moderna homepage)
The government initially announced plans to introduce the Moderna vaccine in the second quarter of this year. Moderna also presented plans to supply it to Korea around May this year. However, due to international vaccine supply shortages, the introduction has faced difficulties. The government plans to additionally introduce 2.71 million doses of Moderna, Janssen, and Novavax vaccines in the first half of the year, but the full-scale introduction of the Moderna vaccine is expected to occur in the second half.
Moderna is highly likely to establish a subsidiary in Korea to pursue vaccine contract manufacturing (CMO). Recently, Moderna posted recruitment notices for employees to work at newly established subsidiaries in Korea, Japan, and Australia. This supports speculation that the 'August domestic CMO-produced vaccine' mentioned by authorities is Moderna. Candidate companies for the CMO contract include GC Green Cross, Hanmi Pharmaceutical, Samsung Biologics, and ST Pharm.
Meanwhile, as of midnight on the same day, the daily new COVID-19 cases in Korea were 463, marking the 400s range for the first time in a week. There were 436 domestic cases and 27 imported cases.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















