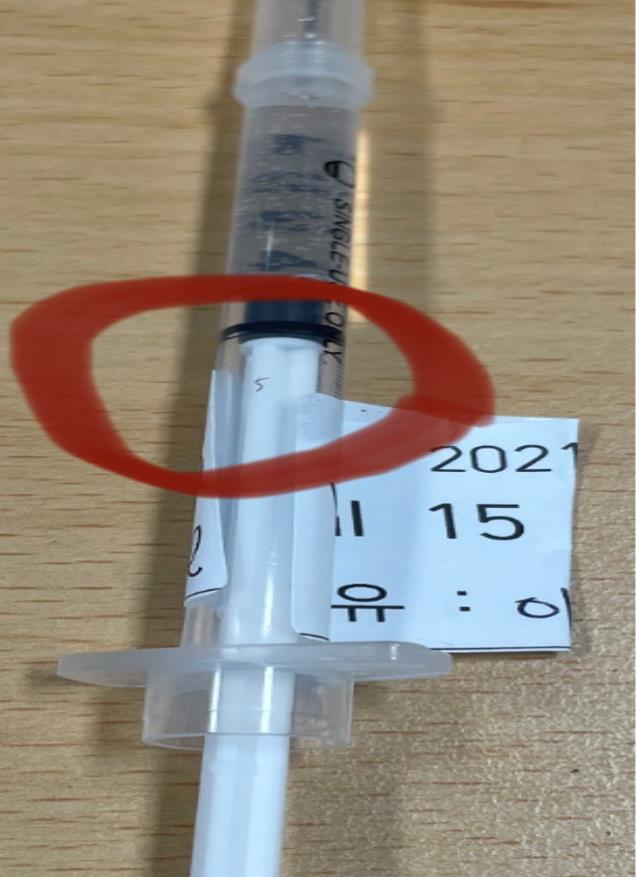
 The Ministry of Food and Drug Safety announced on the 7th that it received three foreign substance reports related to low dead space (LDS) syringes used for COVID-19 vaccinations and ordered corrective and preventive measures from the relevant company. The photo shows an LDS syringe with reported foreign substances. ▲Photo by Ministry of Food and Drug Safety [Image source=Yonhap News]
The Ministry of Food and Drug Safety announced on the 7th that it received three foreign substance reports related to low dead space (LDS) syringes used for COVID-19 vaccinations and ordered corrective and preventive measures from the relevant company. The photo shows an LDS syringe with reported foreign substances. ▲Photo by Ministry of Food and Drug Safety [Image source=Yonhap News]
[Asia Economy Reporter Lee Chun-hee] Foreign substances were found in low dead space (LDS) syringes used for COVID-19 vaccinations, prompting the Ministry of Food and Drug Safety (MFDS) to order cause analysis and corrective and preventive measures.
The MFDS announced on the 7th that it received three reports of foreign substances in LDS syringes manufactured by Company A in Daegu and ordered the company to analyze the cause and implement corrective and preventive actions. On the 4th, two cases were reported where black lines were found on the plunger part of the LDS syringes made by Company A, and one case where black material was found inside the barrel.
However, the MFDS confirmed that in all three cases, the foreign substances did not come into contact with the medication. According to the MFDS, the foreign substances were embedded and fixed in the syringe plunger or barrel parts, making it impossible for them to enter the human body. Additionally, all syringes with foreign substances were discovered during the preparation stage and were not used.
The manufacturer has implemented measures such as improving the post-heat treatment process and increasing quality control personnel to minimize foreign substance contamination.
On the 6th, the MFDS formed a public-private expert consortium including GMP specialists and began efforts to improve product quality by conducting on-site inspections of production facilities and process improvements at four manufacturers supplying LDS syringes.
On the same day, a meeting was held with six domestic syringe manufacturers to analyze the causes of foreign substance contamination in syringes and discuss improvement directions for reduction. During the meeting, it was discussed that parts used in syringes are plastic injection molded products, and carbonized raw material particles (black spots) can be included in the molded parts during the heat treatment process, leading to reports of foreign substances. The meeting concluded that these products are not foreign substances but surface defects, posing no risk of entering the human body or contaminating medication. However, it was agreed to form a government-industry consultative body to closely discuss ways to filter out defective products through more thorough process inspections.
An MFDS official stated, "We have urged syringe manufacturers to thoroughly inspect equipment cleaning and manufacturing facility maintenance to prevent foreign substances or quality defects during the manufacturing process and will continue to provide technical support for quality management."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















