50,000 AZ Vaccines Already Administered
"Low Risk... No Adverse Reactions Reported"
630,000 Units Recalled by the 16th
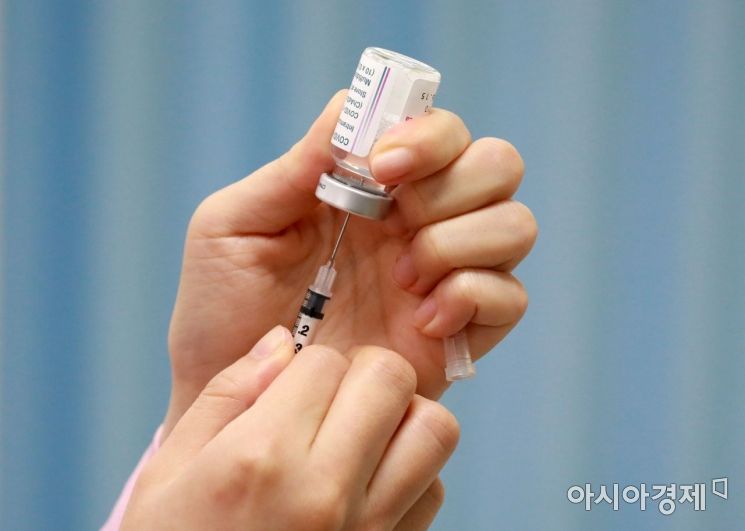 On the 2nd, a medical staff member at Mapo-gu Public Health Center in Seoul is busy filling syringes with the AstraZeneca COVID-19 vaccine ahead of vaccination for health and medical organization leaders. Photo by Hyunmin Kim kimhyun81@
On the 2nd, a medical staff member at Mapo-gu Public Health Center in Seoul is busy filling syringes with the AstraZeneca COVID-19 vaccine ahead of vaccination for health and medical organization leaders. Photo by Hyunmin Kim kimhyun81@
[Asia Economy Reporter Kim Ji-hee] Foreign fiber contaminants were found in 'Low Dead Space (LDS)' syringes used for COVID-19 vaccinations, prompting health authorities to recall 700,000 syringes from vaccination sites.
A Korea Disease Control and Prevention Agency official stated on the 17th, "There have been 21 reports of foreign substances found inside the syringes, and the manufacturer of the LDS syringes is proactively conducting a recall," adding, "The planned recall quantity for this week is 700,000 units." As of the 16th, approximately 630,000 units have already been collected.
Among the 21 reported cases, 19 involved products from DuWon Meditech, while one case each was reported for products from Shin-A Yanghaeng and Poonglim Pharmatech. The syringes currently being recalled are from DuWon Meditech. The report concerning Shin-A Yanghaeng’s product involved foreign substances found in parts other than the medication reservoir, so no quality issues were identified. For Poonglim Pharmatech’s product, no foreign substances were found during the verification process following the report.
Of the DuWon Meditech syringes, 500,000 units have already been used for AstraZeneca (AZ) vaccine administration. However, authorities and experts believe that it is unlikely for the foreign substances to pass through the syringe filter and enter the body. The government stated, "No related adverse reaction cases have been reported so far."
This recall is not due to an official government order but is a proactive measure taken by the companies involved. The companies plan to resume production and supply after improving the LDS syringe manufacturing process and quality in compliance with medical device regulations.
LDS syringes are designed to minimize the space between the piston and needle to reduce residual liquid waste. Using these syringes allows for administering COVID-19 vaccines to one or two additional people per vial.
The first report of foreign substances in LDS syringes was received on February 27 of this year. However, the government only issued a suspension of use three weeks later, on March 18. Reports by region include 5 in Seoul, 6 in Gyeonggi, 1 in Incheon, 3 in Busan, 1 in Chungnam, 3 in Gyeongbuk, and 2 in Gyeongnam.
Regarding criticism over the delayed government announcement after the reports, the Ministry of Food and Drug Safety explained, "After receiving the foreign substance reports, we conducted a cause investigation and corrective orders, and in consultation with the KDCA, issued a suspension of use. Since the companies voluntarily took measures such as exchanges, administrative sanctions or public announcements were not necessary." They added, "We will strengthen quality control of quarantine-related supplies through the technical support team and provide more detailed information where needed."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)










