Maximum production capacity of 100 million doses per month
Long-term domestic utilization potential is positive, but
immediate domestic supply issues remain unresolved
Authorities: "The mentioned August domestic contract vaccine production is not Sputnik V"
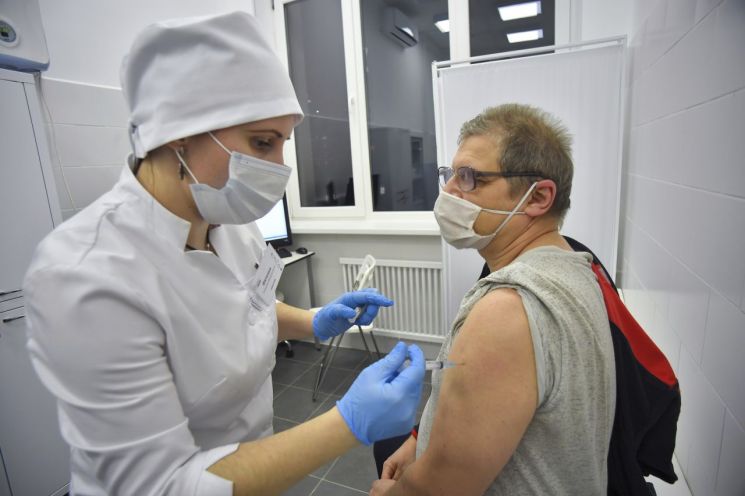 A medical worker in Moscow, Russia, is administering the 'Sputnik V' COVID-19 vaccine to an elderly man. [Image source=AP Yonhap News]
A medical worker in Moscow, Russia, is administering the 'Sputnik V' COVID-19 vaccine to an elderly man. [Image source=AP Yonhap News]
[Asia Economy reporters Seo So-jeong and Lee Chun-hee] Domestic bio company Huons Global is set to undertake contract manufacturing (CMO) of the Russian COVID-19 vaccine 'Sputnik V.' With a maximum production capacity of 100 million doses per month, expectations are rising as a domestic company will directly produce the COVID-19 vaccine through technology transfer.
However, the initial batch is expected to be entirely for overseas export, making it difficult to immediately resolve the domestic vaccine shortage. The government also clarified that the previously mentioned 'domestic contract-produced vaccine in August' is not the Sputnik V vaccine.
Huons Global, the holding company of the Huons Group, announced on the 16th that it has signed a technology transfer agreement with the Russian sovereign wealth fund (RDIF) for the production of the Sputnik V COVID-19 vaccine and will begin pilot production in August.
Huons Global plans to form a consortium with Prestige BioPharma, Humedix, Boran Pharma, and others, mobilizing each company's capabilities to establish facilities capable of producing over 100 million doses per month. A Huons Global official stated, "While facility construction will enable production of up to 100 million doses, actual production volume will be based on the quantity requested by RDIF," adding, "All doses produced this time will be for overseas export."
Separately, domestic bio company Gelra Pharma has formed a consortium centered on its subsidiary Korea Chorus, along with seven domestic institutions and companies, for the production of the Sputnik V vaccine. Although Humedix also participates in this consortium, it is distinct from the Huons Global consortium. The Korea Chorus consortium reportedly has an annual production capacity of 650 million doses.
The Sputnik V vaccine was the world's first COVID-19 preventive vaccine to receive approval in August last year. It is currently authorized for use in over 60 countries worldwide, including Russia, Iran, Argentina, Algeria, and Hungary. The European Medicines Agency (EMA) also began its review earlier this month.
In February, the global medical journal The Lancet published Phase 3 clinical trial results showing a preventive efficacy of 91.6% for Sputnik V. Although early approval by Russian authorities sparked safety concerns, recent Phase 3 results have demonstrated its effectiveness, changing the overall evaluation. In particular, attention is focused on whether domestic companies' involvement in COVID-19 vaccine production will positively impact domestic supply and utilization.
Son Young-rae, head of the Social Strategy Division at the Central Disaster and Safety Countermeasures Headquarters, mentioned The Lancet paper during the regular briefing of the Central Disaster and Safety Countermeasures Headquarters that morning, stating, "Foundations for evaluation have been established, and not only South Korea but the entire world is paying attention as various verifications are currently underway." However, he also expressed a reserved stance, saying, "We need to observe further whether it is an area that can be sufficiently introduced," and "We will consider and watch with interest."
In fact, the Sputnik V vaccine is a viral vector vaccine developed using an adenovirus vector, the same type used in the AstraZeneca and Janssen vaccines, which have been associated with rare thrombosis accompanied by thrombocytopenia, posing a safety variable. Professor Jung Jae-hoon of the Department of Preventive Medicine at Gachon University Gil Medical Center said, "Although the Russian government denied the possibility of thrombosis with the Sputnik V vaccine, verification is necessary given that the same vector was used."
Meanwhile, Baek Young-ha, head of the Vaccine Introduction Task Force's Vaccine Introduction General Team, stated in a briefing the previous day, "A domestic pharmaceutical company is in the process of signing a specific contract for the production of an overseas-approved vaccine," adding, "Mass production of an overseas-approved vaccine is scheduled to begin domestically from August." Following the government's announcement, the contract of the Huons Global consortium was signed the next day, leading to speculation that the vaccine mentioned by the government might be the Sputnik V vaccine. Huons Global's stock price also surged rapidly, recording 51,800 won at 11:36 a.m., up 29.82% from the previous day.
However, Son clarified, "The announcements made yesterday were not related to the Russian Sputnik V vaccine," emphasizing, "They were intended to inform about the government's efforts to create a favorable environment for vaccine supply and to alleviate public anxiety."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















