EMA "Still Under Investigation, Results to be Announced on 7th-8th"
Oxford University Also Halts Pediatric Clinical Trials Completely
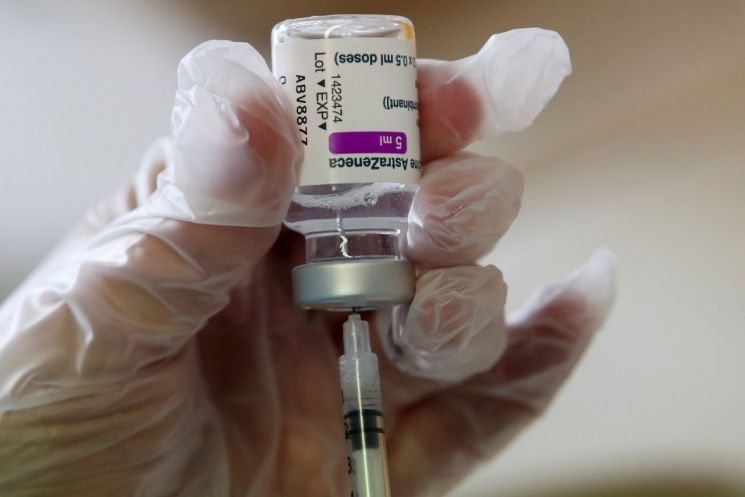
[Asia Economy Reporter Hyunwoo Lee] The head of vaccine strategy at the European Medicines Agency (EMA) has claimed that the COVID-19 vaccine jointly developed by AstraZeneca (AZ) and the University of Oxford in the UK is believed to be causally linked to rare thrombosis side effects observed after vaccination, sparking global repercussions. Although the EMA is still investigating and has refrained from making an official statement, there is speculation that it may reverse its previous position from last month, which stated there was no connection to thrombosis. If a causal relationship between the AZ vaccine, which has already been administered to tens of millions in 94 countries worldwide, and rare thrombosis is confirmed, there are concerns that vaccinations could be halted globally.
According to foreign media including the Associated Press on the 6th (local time), Marco Cavaleri, head of vaccine strategy at EMA, said in an interview with the Italian daily Il Messaggero, "In my personal opinion, it seems clear that there is a causal relationship between the AZ vaccine and side effects such as thrombocytopenia and rare thrombosis," adding, "However, we do not yet know why these reactions occur, and determining this is a key task going forward." He continued, "EMA will soon acknowledge the association between the AZ vaccine and rare thrombosis," but added, "These cases are very rare, and I still believe the benefits of vaccination outweigh the risks of side effects."
Following these remarks and the ensuing widespread controversy over the AZ vaccine, EMA issued an official statement saying, "No conclusion has yet been reached regarding the relationship between the AZ vaccine and rare thrombosis, and a related review is currently underway." It further stated, "Once the review is completed between the 7th and 8th, the investigation results will be disclosed at a press conference."
Accordingly, concerns have been raised that EMA may reverse its earlier announcement that there is no link between AZ vaccination and increased thrombosis risk. Previously, last month, EMA stated that the AZ vaccine does not increase the risk of thrombosis but noted that additional analysis was needed regarding rare occurrences of disseminated intravascular coagulation (DIC) and cerebral venous sinus thrombosis (CVST), which are specific types of thrombosis.
EMA is currently holding a safety committee meeting to evaluate the relationship between the AZ vaccine and rare thrombosis as well as safety thresholds, and it is reported that new vaccination recommendations will be issued during the plenary session continuing until the 9th.
On the same day, the University of Oxford, which co-developed the vaccine with AZ, announced that it will fully suspend ongoing pediatric clinical trials of the AZ vaccine for children and adolescents until the EMA’s investigation results are finalized and announced. This comes amid growing safety concerns following the announcement by the UK Medicines and Healthcare products Regulatory Agency (MHRA) the previous day that it is considering halting AZ vaccinations for people under 30 years old.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















