[Asia Economy Reporter Lee Chun-hee] Celltrion's COVID-19 antibody treatment 'Rekkirona (CT-P59)' has begun the review process for emergency use prior to formal approval in Europe.
On the 2nd (local time), Celltrion announced on the 3rd that the European Medicines Agency (EMA) has initiated a review process to provide expert opinions to European countries seeking early introduction of Rekkirona.
This means that experts from the Committee for Medicinal Products for Human Use (CHMP) under the EMA will provide scientific opinions on the availability of Rekkirona so that individual European national health authorities, urgently needing Rekkirona, can make introduction decisions through 'Compassionate Use Programs' before EMA's formal marketing authorization.
This review is based on Celltrion's clinical data conducted on COVID-19 patients with moderate symptoms and high-risk groups at risk of progressing to hospitalization. EMA will verify whether Rekkirona can reduce the rate of progression to severe COVID-19 and decrease visits and admissions to intensive care units.
This procedure is separate from the rolling review initiated on the 24th of last month. Previously, Celltrion submitted quality and manufacturing process management, non-clinical test, and clinical trial data to the EMA for the rapid marketing authorization of Rekkirona through this process. Accordingly, CHMP has begun evaluating the efficacy and safety of Rekkirona based on the various data submitted by Celltrion.
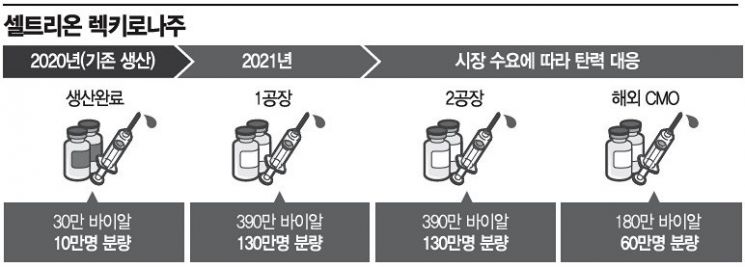
Celltrion is also smoothly securing supply quantities immediately after completing the global rapid approval process. Production for 100,000 doses has already been completed. Celltrion plans to additionally produce 1.5 million to 3 million doses of Rekkirona annually according to demand to respond promptly to supply needs.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















