UNIST Professor Jangbeom Baek's Team Synthesizes 2D Organic Semiconductor Material with Tens of Times Improved Charge Mobility
Exhibits Excellent Electrical Conductivity upon Hydrogen Chloride Doping … Published in Advanced Materials
 Film-type synthesis photo of the material developed in the research that dramatically improved the charge mobility efficiency of organic semiconductors.
Film-type synthesis photo of the material developed in the research that dramatically improved the charge mobility efficiency of organic semiconductors.
[Asia Economy Yeongnam Reporting Headquarters Reporter Kim Yong-woo] A study that has raised the charge mobility of ‘organic semiconductors’ used in rollable displays and wearable electronic devices to the highest level has attracted attention.
Organic semiconductors are excellent materials in terms of flexibility, but their charge mobility is low, limiting their applications. This study has opened new possibilities.
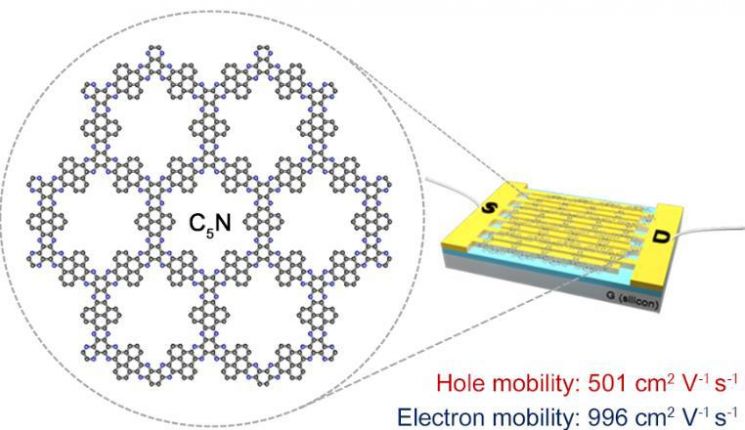
The research team led by Professor Baek Jong-beom of the Department of Energy and Chemical Engineering at Ulsan National Institute of Science and Technology (UNIST, President Lee Yong-hoon) succeeded in synthesizing a ‘C5N (C-five-N) two-dimensional organic polymer structure’ through an aromatic cyclization reaction.
When this organic polymer structure was made into a thin film and used in a semiconductor transistor device (FET), the charge mobility increased by more than several tens of times. Furthermore, doping this structure with hydrochloric acid (HCl) significantly enhanced its electrical conductivity, enabling its use as a conductive material.
The research team obtained the C5N (C-five-N) structure by reacting two types of chemicals (HAB and PTK). Unlike graphene, which consists of hexagonal rings made only of carbon (C), this two-dimensional structure has uniform pores and nitrogen atoms (N) added, resulting in excellent charge mobility (electron 996 cm²V?¹s?¹, hole 501 cm²V?¹s?¹).
This is the highest charge mobility reported so far for organic semiconductors. Charge mobility refers to the speed at which electrons or holes move within a material. Semiconductor devices made from materials with low charge mobility suffer from slow electrical signal transmission, causing issues such as color conversion delays in displays.
The ring structure of the two-dimensional material developed by the research team was obtained through an ‘aromatic cyclization reaction,’ making it very stable and capable of withstanding high temperatures up to 600°C.
Javeed Mahmood, Ph.D., the first author, explained, “Since every part of the structure is ring-shaped, it enhances chemical and thermal stability compared to existing two-dimensional organic structures,” adding, “It can be used under various high-temperature conditions.”
Moreover, this structure exhibits superior electrical conductivity compared to conventional conductive polymers like chain-type polyaniline. When doped with hydrochloric acid (HCl), its conductivity improves more than 140 times (1038 S/cm), making it usable as a versatile conductive polymer.
Organic semiconductors have been actively researched over the past few decades as materials to replace inorganic silicon semiconductors due to their flexibility, light weight, low processing costs, and ease of property control.
However, most organic semiconductors have struggled to replace inorganic semiconductor materials due to their lower-than-expected charge mobility.
Professor Baek Jong-beom, the lead researcher of this study, said, “This research has overcome the chronic problem of low charge mobility when using two-dimensional polymers as organic semiconductor materials,” and added, “We expect significant progress in the development of organic semiconductor devices in the future.”
This study, conducted in collaboration with Professor Oh Jun-hak’s research team from the Department of Chemical and Biological Engineering at Seoul National University, was published in the prestigious international journal Advanced Materials on January 20.
The research was supported by the Ministry of Science and ICT’s Leader Researcher Support Project (Creative Research), BK21 Plus Project, Excellent Science Research Center (SRC), and U-K Brand Development Project (UNIST).
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.