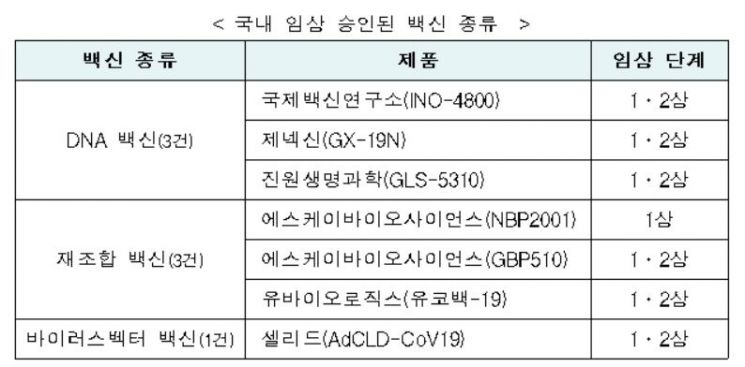
The Number of Vaccines in Domestic Clinical Trials Increases to Seven
[Asia Economy Reporter Kim Ji-hee] Clinical trials for a novel coronavirus disease (COVID-19) vaccine developed by UbioLogics are set to begin.
On the 21st, the Ministry of Food and Drug Safety announced that it approved UbioLogics' COVID-19 vaccine "Ucovac-19" clinical trial plan on the 20th. With this, there are currently 7 vaccine products and 15 therapeutic products in development in South Korea that have received clinical trial plan approval related to COVID-19.
This clinical trial aims to evaluate the safety and immunogenicity of Ucovac-19 in healthy adults. Phase 1 will be conducted first, followed sequentially by Phase 2. Ucovac-19 is a "recombinant vaccine" created using genetic recombination technology to produce the surface antigen protein of the COVID-19 virus. The vaccine’s surface antigen protein stimulates immune cells to form neutralizing antibodies, inducing an immune response, and when the COVID-19 virus invades, the antibodies eliminate it.
Ucovac-19 uses liposomes as an adjuvant ingredient to enhance the vaccine’s immune response and consequent clinical efficacy. The surface antigen protein is expressed on the surface of the liposomes to induce an immune response. Overseas, companies such as Novavax in the United States (Phase 3) are also conducting clinical trials of COVID-19 vaccines using genetic recombination technology.
The Ministry of Food and Drug Safety stated, "Considering the high public interest in the development of COVID-19 therapeutics and vaccines, we will promptly provide information on the status of clinical trials for development products," and added, "We will do our best to support the rapid development of safe and effective COVID-19 therapeutics and vaccines so that our citizens can be guaranteed treatment opportunities."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.