"Review Planned After WHO Clinical Trial Final Results Announcement"
[Asia Economy Reporter Seo So-jeong] The Ministry of Food and Drug Safety (Minister Lee Eui-kyung) and the Korea Disease Control and Prevention Agency (Director Jung Eun-kyung) announced on the 23rd that the administration of Remdesivir to severe COVID-19 patients under medical judgment continues to be recommended.
Recently, the World Health Organization (WHO) announced that the clinical trial results of Remdesivir showed no difference in mortality and treatment duration between the control group and the experimental group. Since the study results have not yet undergone peer review in the academic publication process, the Ministry of Food and Drug Safety plans to review related matters once the final results of the WHO clinical trial are released.
Based on expert consultation, the Ministry of Food and Drug Safety and the Korea Disease Control and Prevention Agency determined that additional data review is necessary, including analysis by timing of administration and subgroups by severity.
Once the final results of the WHO clinical trial are published, a thorough review of the enrolled patients, the regional medical environment where the trial was conducted, and the trial methods and results will be conducted. Discussions with experts and overseas regulatory authorities will also be held.
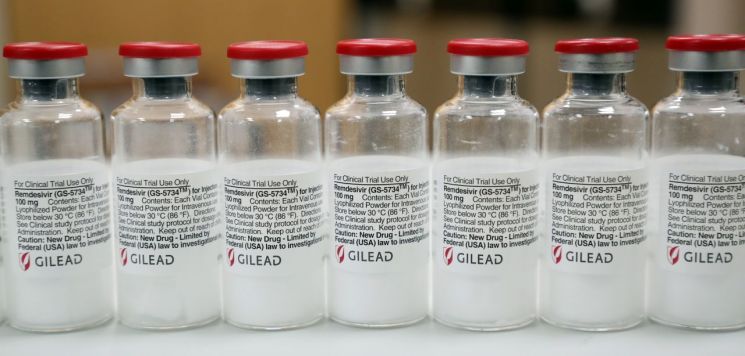
The Ministry of Food and Drug Safety approved the use of Remdesivir on July 24 for severe hospitalized patients requiring supplemental oxygen, based on clinical trial results led by the U.S. National Institute of Allergy and Infectious Diseases (NIAID) for the treatment of domestic COVID-19 patients.
The Ministry of Food and Drug Safety judged that the result showing Remdesivir shortened the treatment period by 5 days in COVID-19 patients is clinically meaningful. Remdesivir has been approved and is in use in the United States, Europe, Japan, Taiwan, Singapore, and other countries.
The Ministry of Food and Drug Safety’s COVID-19 Expert Committee and the Central Clinical Committee on Emerging Infectious Diseases stated that the U.S. NIAID used highly reliable research methods and that there is scientific evidence of the drug’s efficacy, thus maintaining the position that Remdesivir should continue to be used as a treatment for COVID-19.
The Ministry of Food and Drug Safety and the Korea Disease Control and Prevention Agency said, "We will continue to monitor clinical trials and adverse events related to COVID-19 treatments and vaccines, and do our best to introduce safe and effective medicines domestically to ensure that our citizens have access to treatment opportunities."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















