AstraZeneca Clinical Success in UK... "Possible UK Launch in September, US in October"
Pfizer Also Succeeds in Phase 2, Sinovac Enters Phase 3... Vaccine Production Expected by Early Next Year
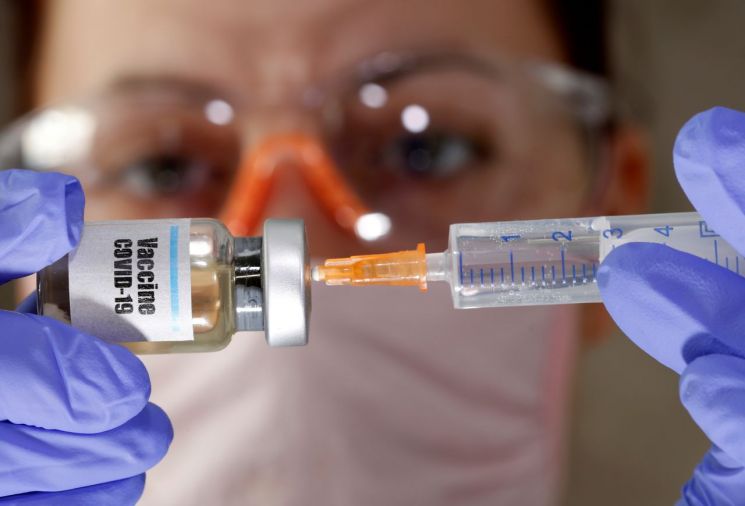
[Asia Economy Reporter Hyunwoo Lee] British pharmaceutical company AstraZeneca, which is developing a vaccine for the novel coronavirus infection (COVID-19), has confirmed antibody formation in a clinical trial involving more than 1,000 participants. Previously, American pharmaceutical company Moderna announced that antibodies were confirmed in all participants in its Phase 1 trial, but it was not conducted on such a large scale as AstraZeneca's. AstraZeneca is expected to be the first in the world to launch a vaccine as early as September. American company Pfizer also announced success in its Phase 2 trial on the same day. As pharmaceutical companies continue to announce clinical trial successes, expectations for vaccine release within the year are growing.
According to foreign media including CNBC on the 20th (local time), British pharmaceutical company AstraZeneca announced that in the Phase 1 trial of the COVID-19 vaccine, neutralizing antibodies and T cells were formed in all 1,077 participants. Neutralizing antibodies and T cells are known to be antibodies and cells that destroy viruses. Adrian Hill, a researcher at Oxford University, stated, "Considering AstraZeneca's manufacturing capabilities, it will be possible to produce one million doses of the vaccine by September." AstraZeneca has completed Phase 2 trials and began Phase 3 trials earlier this month. The company is also a beneficiary of the U.S. government's 'Operation Warp Speed,' which funds companies developing COVID-19 vaccines. It is expected that vaccine supply will begin in September in the UK and in October in the U.S.
Following the announcement of the Phase 1 trial success, AstraZeneca's stock price rose. On the London Stock Exchange that day, the company's stock closed at 93.20 pounds (approximately 140,000 won), up 1.45% from the previous day. The stock price surged more than 10% in early trading but the increase was significantly reduced due to growing external risk concerns, such as the UK government's announcement to suspend the extradition treaty with Hong Kong.
American company Pfizer also announced clinical trial success on the same day. According to the Associated Press, Pfizer announced that neutralizing antibodies and T cells were formed in the Phase 2 trial of the COVID-19 vaccine jointly developed with German biotech company BioNTech. Phase 3 trials will begin at the end of this month, with plans to start vaccine production early next year.
Chinese pharmaceutical company Sinovac is also starting Phase 3 clinical trials for its COVID-19 vaccine. The government of S?o Paulo state in Brazil announced at a press conference that "Phase 3 clinical trials using Sinovac's vaccine will begin on the 21st at hospitals in S?o Paulo city." The Butantan Institute in Brazil, which is collaborating on vaccine research with Sinovac, mentioned that 9,000 people nationwide will participate in this clinical trial, which is expected to conclude after 90 days. They added that vaccine production will begin early next year.
The spread of COVID-19 remains strong worldwide, including in the United States. According to the statistics site Worldometer, 205,348 new confirmed cases were added worldwide on the 20th alone. As of that day, the cumulative confirmed cases totaled 14,844,358, with 612,795 deaths recorded. The United States, which has suffered the most, is approaching 4 million cumulative confirmed cases.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)










