Large-Scale Recruitment in Production and Quality
110 Billion Won Invested in Songdo Plant Expansion
STgen Bio, a contract manufacturing organization (CMO) subsidiary specializing in biopharmaceuticals under Dong-A Socio Holdings, has embarked on a large-scale expansion of its facilities and workforce. The company is accelerating its external growth in line with the increase in commercial production volumes.
According to the pharmaceutical and biotechnology industry on February 27, STgen Bio is currently conducting large-scale recruitment, focusing on production and quality divisions. Starting in April, the company will overhaul its on-site operation system, shifting from the current four-team, three-shift schedule to a four-team, two-shift schedule. A company representative stated, "Recently, the rapid increase in commercial production volumes has intensified the workload for on-site employees," adding, "We are reorganizing the shift system and strengthening our workforce to ensure stable production line operation and efficient management."
The company is also making aggressive facility investments. STgen Bio is investing a total of 110 billion won to expand its first plant in Songdo, Incheon. The expansion plan includes the addition of two 2,500-liter bioreactors, one harvest tank, and an isolator filling line. Once the expansion is complete, STgen Bio’s production capacity will increase from the current annual 9,000 liters to 14,000 liters. The company also aims to boost its finished drug product (DP) manufacturing capacity by more than 170% compared to the current level.
The driving force behind STgen Bio’s steep growth trajectory is attributed to the global supply expansion of 'IMULDOSA,' a Stelara biosimilar developed by affiliate Dong-A ST. The company’s achievement last year of both cGMP approval from the U.S. Food and Drug Administration (FDA) and EU-GMP certification from the European Medicines Agency (EMA) proved its global-level quality capabilities, which played a key role. This milestone has reportedly led to more active contract negotiations with major global pharmaceutical companies.
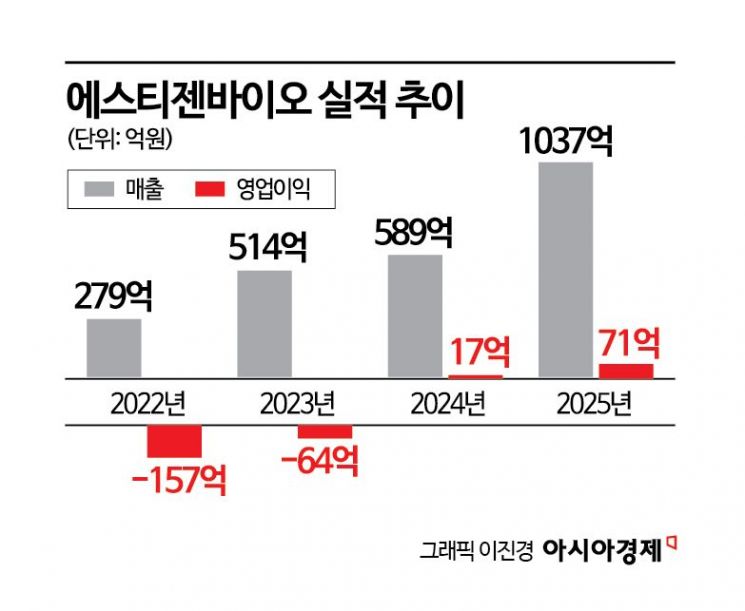
The company’s performance is already on an upward curve. Last year, STgen Bio posted sales of 10.37 billion won, representing a 76.2% increase year-on-year, and operating profit of 7.1 billion won, a surge of 308.6%. Once STgen Bio achieves a 14,000-liter manufacturing system, it is expected to solidify its position as a major player in the domestic small- and mid-sized CDMO market alongside companies like Binex (which is expected to reach around 22,000 liters). STgen Bio emphasizes its unique strength as the only company in Korea capable of producing both DS and DP at a single site with GMP capabilities.
Industry attention is now turning to the timing of STgen Bio’s initial public offering (IPO). The appointment late last year of Hyunmin Lee, a 'finance expert' and former head of planning at Dong-A Socio Holdings, as CEO has added weight to this speculation. With the transition to the commercialization phase and the leadership of financial and management professionals-at a time when the reappointment of former CEO Kyungeun Choi, a quality expert who led cGMP certification, was also under consideration-the market interprets these moves as preparations for an IPO and other capital market activities. An industry insider commented, "If additional orders to fill the expanded capacity become visible, the IPO process will gain even more momentum."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.