Yuhan Corporation's Obesity Drug Candidate Draws Attention
GDF15-Brain Appetite Circuit Identified in Cancer Cachexia Research
Competition Heats Up for Central Nervous System-Targeted Obesity Treatments After GLP-1
As key evidence emerges regarding next-generation obesity treatments that directly regulate appetite signals in the brain, research into new targets following glucagon-like peptide-1 (GLP-1) is expected to gain traction. While GLP-1-based therapies have indirectly suppressed appetite via the gut-brain axis, approaches that regulate the central nervous system's appetite circuits themselves are emerging as a new paradigm.
According to the medical community on February 26, a research team at the University of Oklahoma in the United States has identified a "triangular regulation" mechanism involving interactions among tumor cells, immune cells, and the central nervous system. The team recently published their findings in the international journal 'Cancer Cell.'
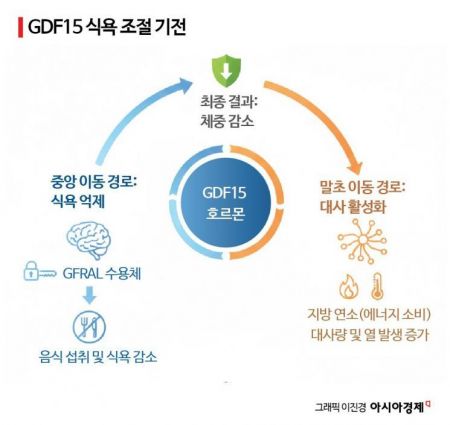
This study investigated how cancer alters the body's overall appetite regulation system. The researchers uncovered the process by which tumor cells stimulate immune cells and the brain, leading to increased secretion of the stress-responsive cytokine GDF15. They demonstrated in disease models that GDF15 is a key mediator of central appetite control, providing a mechanistic foundation for the development of obesity treatments targeting GDF15.
According to the study, the identified "triangular regulation circuit" has been pointed out as a major cause of cachexia and loss of appetite frequently observed in cancer patients. Cachexia refers to a condition in which patients with cancer or other serious illnesses experience rapid loss of muscle and body fat, along with severe appetite decline. The research confirmed that weight loss in cachexia patients is not simply due to metabolic abnormalities in the body, but rather results from changes in appetite regulation signals caused by direct stimulation of GDF15 receptors in the brain.
Recently, GLP-1-based obesity treatments have been drawing explosive popularity by mimicking the action of gut-secreted hormones to deliver appetite-suppressing signals to the brain indirectly. In contrast, some academic circles believe that directly targeting appetite regulation signals generated in the brain itself could result in a more powerful weight reduction effect. As research clarifying the relevant mechanisms continues to be published, new drug candidates aiming at targets different from GLP-1 are drawing attention as latecomers in the obesity treatment field.
In Korea, Yuhan Corporation is developing an obesity treatment that directly regulates brain appetite signals using a GDF15 variant-based candidate compound. Unlike existing incretin-based drugs, which indirectly suppress appetite through the gut-brain axis, this approach directly targets central nervous system appetite regulation pathways and is being evaluated as a next-generation strategy. A Yuhan Corporation representative stated, "We are currently preparing to enter preclinical trials."
Among global pharmaceutical companies, there is a case where Novartis attempted to develop an obesity treatment targeting the GDF15 signaling axis but halted the effort. Novartis developed the candidate compound 'MBL949,' which activates the GDF15 receptor pathway, as an obesity treatment, but removed it from their pipeline after it failed to deliver the expected weight loss effect in Phase 2 clinical trials. The Novartis study is seen as an example that, while directly regulating central appetite signals holds great potential, there may be limitations to controlling obesity through a single pathway alone.
Efforts to explore new mechanisms beyond GLP-1 are spreading to other Korean companies as well. Hanmi Pharmaceutical is developing a non-incretin-based candidate compound targeting the 'CRF2 receptor,' which is involved in stress hormone signaling, and is pursuing a strategy aimed at both reducing fat and preserving muscle mass. This approach not only suppresses appetite but also seeks to alter the metabolic regulation axis itself, differentiating it from existing GLP-1-based therapies.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.