A New Synthesis Technology for Precious Metal Single-Atom Catalysts
Achieved Through High-Temperature Self-Assembly-Assisted Dynamic Placement Strategy
Korean researchers have succeeded in improving the economic feasibility of water electrolysis cells by reducing the amount of platinum used to one-tenth.
On the 31st, the research teams of Professor Jinwoo Lee from the Department of Biological and Chemical Engineering at KAIST and Professor Hyungjun Kim from the Department of Chemistry announced that through joint research, they developed a high-performance, high-stability precious metal single-atom catalyst that dramatically enhances the performance and stability of anion exchange membrane-based water electrolysis cells.
 Number of research participants and cover paper illustration. From the top of the photo clockwise: Research Professor Seongbin Kim, Professor Seungjae Shin, Dr. Hoyoung Kim, Professor Jinwoo Lee, Professor Hyeongjun Kim. Provided by KAIST
Number of research participants and cover paper illustration. From the top of the photo clockwise: Research Professor Seongbin Kim, Professor Seungjae Shin, Dr. Hoyoung Kim, Professor Jinwoo Lee, Professor Hyeongjun Kim. Provided by KAIST
Water electrolysis cells are a technology that produces hydrogen by electrochemically splitting water, and while they are essential energy conversion technology for the carbon-neutral era, industrial application has required the use of large amounts of expensive platinum.
The research team developed a "self-assembly-assisted precious metal dynamic placement" strategy that exploits the degradation mechanism of precious metal catalysts in reverse. This method is a synthesis technology in which precious metals spontaneously and selectively decompose into single atoms on a carbide support at high temperatures above 1000℃, allowing stable anchoring. Using this technology, they achieved higher performance and stability with only one-tenth the amount of platinum compared to commercial platinum catalysts.
Single-atom catalysts have metal atoms isolated on the surface of supports, exhibiting high catalytic efficiency per precious metal atom, but conventional low-temperature reduction methods had limitations in securing catalyst performance and stability.
The research team proposed a new single-atom catalyst synthesis mechanism called self-assembly-assisted precious metal dynamic placement by applying molecular interactions between precious metal precursors and polymers, as well as interactions between precious metals and supports. Through this, they demonstrated that this technology is applicable not only to platinum but also to various precious metal single-atom catalysts such as iridium, palladium, and rhodium.
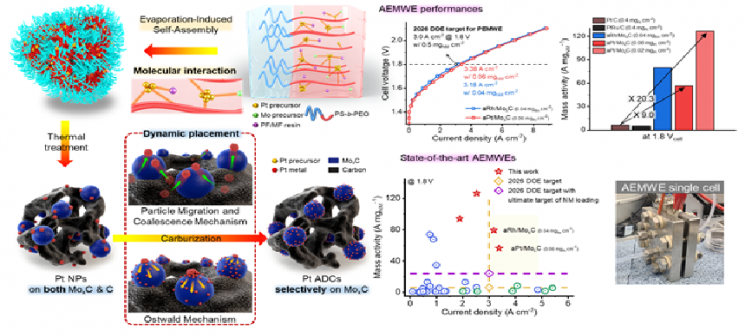 A diagram explaining the principle of the self-assembly original precious metal dynamic placement strategy and the performance of a platinum single-atom catalyst anion exchange membrane-based water electrolysis cell. Provided by the joint research team of Professor Lee Jin-woo at KAIST.
A diagram explaining the principle of the self-assembly original precious metal dynamic placement strategy and the performance of a platinum single-atom catalyst anion exchange membrane-based water electrolysis cell. Provided by the joint research team of Professor Lee Jin-woo at KAIST.
The developed platinum single-atom catalyst showed high stability in alkaline hydrogen evolution reactions and excellent hydrogen production performance through a high density of precious metal active sites, achieving five times higher hydrogen production performance per precious metal atom compared to commercial platinum catalysts.
The water electrolysis cell performance measured in this study was evaluated as the only one to meet the 2026 targets for water electrolysis cell performance and precious metal usage set by the U.S. Department of Energy (DOE).
Research Professor Seongbin Kim, the first author, said, "This technology significantly reduces the cost of water electrolysis cells, and the self-assembly-assisted precious metal dynamic placement strategy presented in this study can be applied not only to water electrolysis cells but also to various precious metal-based catalytic processes, indicating a large industrial impact."
Led by Research Professor Seongbin Kim of the Department of Biological and Chemical Engineering at KAIST, with joint first authorship by Professor Seungjae Shin of the Department of Energy Chemical Engineering at UNIST and Dr. Hoyoung Kim of the Hydrogen Fuel Cell Center at KIST, this study was published in the January issue, volume 18, of the international journal Energy & Environmental Science and was selected as the inside back cover paper.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)









