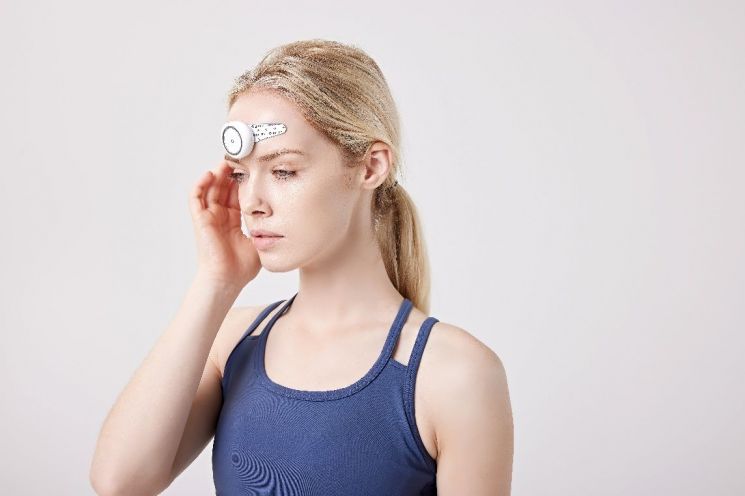
Electronic medicine platform company Ybrain announced on the 20th that its migraine electronic medicine Dupang has obtained 510k approval from the U.S. Food and Drug Administration (FDA). The 510k is a system in which the FDA verifies and approves the safety and effectiveness of medical devices. It had previously received approval from the Ministry of Food and Drug Safety in Korea in 2020.
Dupang works by delivering transcutaneous electrical nerve stimulation (TENS) to the trigeminal nerve area on the forehead that causes migraines, calming the overactive nerves to relieve migraines.
Lee Gi-won, CEO of Ybrain, said, “Through this FDA approval, we have been able to internationally certify the safety and effectiveness of electronic medicine developed with domestic technology.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)