As the patent expiration of the blockbuster ophthalmic disease treatment drug Aylea (generic name Aflibercept) approaches, the pharmaceutical and bio industries are accelerating the approval and clinical trials of biosimilars.
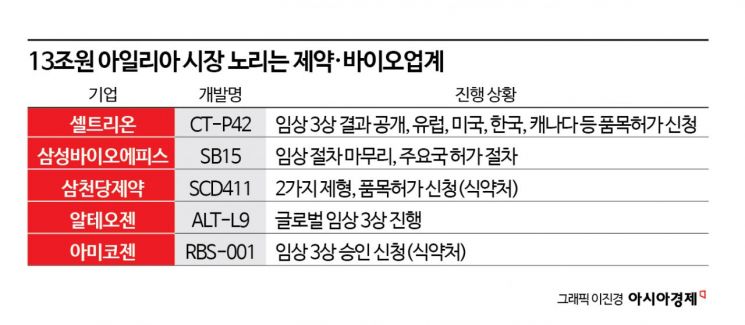
On the 4th, Celltrion disclosed the Phase 3 clinical trial results of the Aylea biosimilar ‘CT-P42’ at the European Society of Retina Specialists. The global Phase 3 trial, conducted over 24 weeks with 348 patients from 13 countries, showed that CT-P42 was similar to the original Aylea not only in the primary endpoint of best corrected visual acuity (BCVA) but also in safety and immunogenicity. Earlier, on the 23rd of last month (local time), Celltrion applied for marketing authorization of CT-P42 to the European Medicines Agency (EMA). Celltrion has also applied for marketing authorization of CT-P42 in the United States (June), Korea (July), and Canada (July), positioning itself as the leading domestic company developing Aylea biosimilars.
Samsung Bioepis also completed the clinical procedures for ‘SB15’ in March last year and is currently undergoing approval processes in major global countries. In April last year, the company released Phase 3 clinical trial results that met equivalence criteria compared to the original drug. The company has stated that it will not disclose specific approval progress to prevent strategic exposure to competitors.
Some companies are pursuing formulation differentiation. Samchundang Pharmaceutical applied for domestic marketing authorization of ‘SCD411’ to the Ministry of Food and Drug Safety on the 30th of last month. Samchundang applied for approval of SCD411 in two formulations: vial and prefilled syringe (PFS). Unlike the vial formulation, which requires transferring the drug into a syringe for administration, the PFS is supplied in a syringe form containing the drug, enhancing dosing convenience and reducing contamination risk. According to the company, Samchundang Pharmaceutical is the only domestic company to apply for approval of an Aylea biosimilar in the PFS formulation.
Additionally, Alteogen is conducting global Phase 3 clinical trials for ‘ALT-L9’, and Amicogen’s subsidiary Lopibio applied for Phase 3 clinical trial approval for ‘RBS-001’ to the Ministry of Food and Drug Safety on the 4th.
Aylea, developed by the U.S. pharmaceutical company Regeneron, is a drug used to treat ophthalmic diseases such as macular degeneration and macular edema. The macula, located at the center of the retina, is responsible for over 90% of vision and is a neural tissue where most photoreceptors are concentrated. Macular degeneration refers to the deterioration of the macula. When degeneration occurs, symptoms such as decreased vision, central scotoma where parts of objects are not visible, and metamorphopsia where objects appear distorted, may appear.
Aylea ranks first in global sales among macular degeneration treatments. It is a blockbuster drug that generated sales of approximately $9.76 billion (about 12.7 trillion KRW) last year alone. The global market for macular degeneration treatments, including Aylea, is expected to grow from $8.9 billion (about 11.66 trillion KRW) in 2020 to $15.3 billion (about 20 trillion KRW) by 2027. The compound annual growth rate during this period is projected to be 8.09%. Aylea’s U.S. market exclusivity is set to expire in May next year. The substance patent in Europe will expire in November 2025.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)