FDA Approves Ferring's 'Levyota'
Ceres' 'SER-109' Decision Set for April Next Year
Growing Interest in Korea
Yuhan's 'Atozen', CJ's 'ChunLab' Acquisitions
Gobiolab and Genome & Company Developing Therapeutics
[Asia Economy Reporter Lee Chun-hee] The era of curing diseases with microorganisms has dawned. With the approval of the first-ever microbiome treatment in the United States, microbiome therapeutics are gaining momentum, sparking growing interest domestically as well.
According to industry sources on the 2nd, the U.S. Food and Drug Administration (FDA) approved Swiss company Ferring Pharmaceuticals' treatment for recurrent Clostridioides difficile infection (CDI), 'Rebyota (RBX2660),' on the 30th of last month (local time).
CDI is a disease in which the gut microbiota is disrupted due to antibiotic use, causing an overgrowth of Clostridioides difficile bacteria that leads to severe diarrhea. There is currently no definitive treatment. The condition of gut microbiota is so crucial that fecal microbiota transplantation, which involves transplanting healthy donor feces to restore gut microorganisms, is sometimes used as a treatment. It is estimated that 15,000 to 30,000 people die annually from CDI in the United States.
 Perrigo Pharmaceuticals logo (Photo by Perrigo Pharmaceuticals)
Perrigo Pharmaceuticals logo (Photo by Perrigo Pharmaceuticals)
According to Ferring, Rebyota demonstrated a 'treatment success rate' of 70.6% in clinical trials, preventing CDI recurrence for 8 weeks, which was higher than the 57.5% rate in the placebo group. Earlier in September, the FDA's Vaccines and Related Biological Products Advisory Committee (VRBPAC) also voted 13 to 4 and 12 to 4, respectively, that Rebyota is effective and safe for treating recurrent CDI.
Currently, Seres Therapeutics has also submitted a Biologics License Application (BLA) for its CDI treatment 'SER-109.' The Prescription Drug User Fee Act (PDUFA) review deadline is set for April 26 next year. In Phase 3 clinical trials, SER-109 reduced CDI recurrence rates to 8.7% at 8 weeks and 13.7% at 24 weeks, compared to 60% and 53% in the placebo group, respectively.
Microbiome is a portmanteau of 'micro' (microorganism) and 'biome' (ecosystem). It refers to the trillions of microorganisms and their genes residing in the human body. It is known that a 70 kg adult carries about 38 trillion microorganisms inside. Research has revealed that various diseases such as psoriasis, gastroesophageal reflux disease, obesity, colitis, and cardiovascular diseases are linked to individual microbiomes, leading to new approaches to cure diseases through microbiome modulation. Market research firm MarketsandMarkets forecasts the global microbiome market to grow from $269 million (approximately 351.6 billion KRW) next year to $1.37 billion (approximately 1.79 trillion KRW) by 2029.
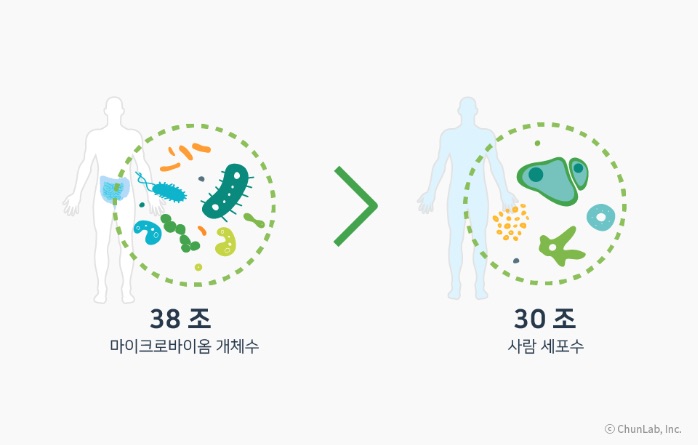 Comparison of the Number of Cells in the Human Body and the Number of Microbiome Organisms (Photo by CJ Bioscience)
Comparison of the Number of Cells in the Human Body and the Number of Microbiome Organisms (Photo by CJ Bioscience)
Domestically, large corporations and major pharmaceutical and bio companies are increasingly acquiring microbiome-related firms, viewing this field as a future growth engine. In September, Yuhan Corporation acquired microbiome therapeutic developer AtoGen from TonyMoly. Subsequently, last month, clinical Phase 1 trials for the bacterial vaginosis treatment 'LABTHERA-001' began in Australia. CJ CheilJedang, which seemed to have exited the bio business after selling CJ Healthcare (now HK Innoen), re-entered the bio sector last year by acquiring ChunLab (now CJ Bioscience).
In Korea, companies such as Gobio Lab, Genome & Company, and CJ Bioscience are developing microbiome new drugs. Research is being conducted on various indications including digestive diseases, cancer, psoriasis, and Alzheimer's disease.
Gobio Lab is leading the field. It is conducting Phase 2 clinical trials for 'KBL697' as a psoriasis treatment and has also submitted an IND (Investigational New Drug application) to the FDA for a Phase 2a clinical trial for ulcerative colitis.
Genome & Company is developing 'GEN-001' as an immuno-oncology agent. It is conducting Phase 2 trials for gastric cancer domestically and recently received approval last month for a Phase 2 combination trial with MSD's (Merck & Co., USA) 'Keytruda' targeting cholangiocarcinoma. CJ Bioscience is also developing anticancer drugs such as 'CLCC1' and 'CJRB-101.'
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)