[DTx Era①] Treating Dementia, ADHD, and Drug Addiction with
Apps, Games, VR, Chatbots, and AI
Amgen, Merck Lead Early Market Entry
Domestic Evaluation Standards Established, Development Underway
Ms. Kim Miseon (pseudonym), who usually suffered from unexplained anxiety and depression that severely disrupted her sleep, eventually found daily life difficult due to lack of sleep and sought medical treatment. Instead of a prescription for medication, the doctor provided her with an access code to download an application (app) on her smartphone. When she opened the app, a treatment program tailored to her symptoms was activated. Following the guidance, Ms. Kim developed habits such as moving her body during the day and meditating before bedtime to maintain a consistent sleep schedule. Without being prescribed oral sleep aids, she was able to change her lifestyle and treat her insomnia according to the doctor's customized prescription.
Digital therapeutics (DTx) refer to software that is not a drug but can treat diseases and improve health like pharmaceuticals. They are utilized in various forms such as mobile apps, games, virtual reality, chatbots, and artificial intelligence. Because of this, they are classified as the third generation of therapeutics, following first-generation synthetic drugs and second-generation biopharmaceuticals. The Digital Therapeutics Alliance (DTA) defines DTx as "highly sophisticated software programs that provide evidence-based therapeutic interventions to patients to prevent, manage, or treat a disease."
Treatment with Smartphone Games Instead of Medicine
DTx targeting specific diseases began in September 2017 when the U.S. Food and Drug Administration (FDA) approved Pear Therapeutics' mobile app for drug addiction treatment called 'reSET.' Pear Therapeutics subsequently received approval for 'reSET-O,' an app for opioid addiction treatment, and 'Somryst,' a DTx for insomnia, holding the most FDA-approved DTx with three products to date.
In November 2017, France's Voluntis received FDA approval for 'Insulia,' an app that calculates insulin doses for type 2 diabetes, and Proteus Digital Health's 'Abilify Mycite,' a treatment for schizophrenia, mania, bipolar disorder, and depression. In 2018, Palo Alto Health Sciences released 'FreeSpira,' an app for treating post-traumatic stress disorder (PTSD) symptoms, followed by Voluntis' 'Oleena' app in 2019, which helps patients self-manage symptoms related to cancer treatment.
In 2020, Akili Interactive launched 'EndeavorRx,' a game designed to treat attention deficit hyperactivity disorder (ADHD) in children aged 8 to 12. It was the first FDA-approved game-based DTx. NightWare developed a program linked to the Apple Watch for people suffering from sleep disorders due to PTSD, and Mahana Therapeutics received approval for 'Gegul8,' a cognitive behavioral therapy digital app for irritable bowel syndrome treatment.
The therapeutic effects of these DTx have also been proven. According to the international pharmaceutical media PharmaForum, a study of 64 patients using reSET-O for 12 weeks by Pear Therapeutics showed a 46% shorter hospitalization period compared to the control group. Consequently, the average hospitalization cost for patients using DTx was $2,693 (approximately 3.47 million KRW), whereas for those not using DTx, it was $6,130 (approximately 7.9 million KRW), more than twice as high.
Participation of Global Pharmaceutical Companies
Leading global pharmaceutical companies have also jumped into the field. U.S. pharmaceutical companies Amgen and Merck invested in Akili Interactive, the creator of the world's first game-based DTx, and Sandoz, a subsidiary of Swiss Novartis, collaborated on the launch of Pear Therapeutics' reSET and reSET-O. They have recognized the potential of the DTx market.
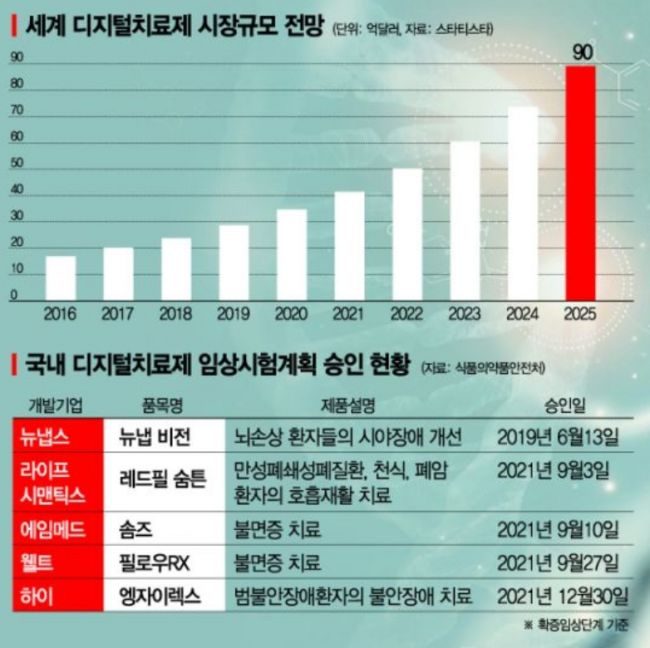
Although the DTx market is still in its early stages, it is growing rapidly in scale. According to Allied Market Research, a U.S. market research firm, the global DTx market size is expected to grow from $2.12 billion (approximately 2.4 trillion KRW) in 2018 to $9.64 billion (approximately 11 trillion KRW) by 2026, with an average annual growth rate of 19.9%. The market is projected to expand to $23.569 billion (approximately 30.34 trillion KRW) by 2030. Euromonitor International estimates the DTx market will reach $8.7 billion by 2025.
In South Korea, the DTx industry is still in its infancy, making it premature to discuss market size. However, the Ministry of Food and Drug Safety established guidelines for the approval and review of digital therapeutic devices in August 2020 and has set evaluation criteria for digital therapeutic devices by various symptoms to support product commercialization.
Although related regulations are still being developed, many companies have already entered the market, so visible results are expected soon. Traditional pharmaceutical and bio companies such as SK Biopharm, Handok, and Samjin Pharmaceutical have also started developing DTx either independently or through equity investments.
Kang Young-gyu, head of the Digital Health Regulatory Support Division at the Ministry of Food and Drug Safety, said, "DTx must also prove safety and efficacy through clinical trials and evidence-based literature targeting patients. It must be clearly presented which software functions are applied to treatment and how feedback and therapy proceed to be managed within the regulatory framework as medical devices."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)