Daegu Gyeongbuk Institute of Science and Technology Research Team

[Asia Economy Reporter Kim Bong-su] Domestic researchers have developed a stroke treatment that maximizes efficacy while minimizing side effects to reduce mortality.
Daegu Gyeongbuk Institute of Science and Technology (DGIST) announced on the 28th that Professor Moon Jae-il of the Department of Brain Science, in collaboration with Professor Lee Sung-yong of Keimyung University School of Medicine, developed a stroke treatment candidate by creating a peptide derivative that modifies the structure of the biological hormone erythropoietin (EPO) based on molecular modeling, minimizing side effects.
Stroke is a disease in which blood vessels supplying blood to the brain are blocked (ischemic stroke) or burst (hemorrhagic stroke), leading to death or physical disabilities caused by brain damage. According to the 2018 death statistics from the National Health Insurance Service, stroke ranks fourth among all causes of death in Korea and is the leading cause of death among single diseases. Once it occurs, even if survival is possible, it is a severe disease accompanied by serious neurological dysfunction.
Erythropoietin (EPO) is a hormone involved in red blood cell production and shows cell-protective effects in various tissues including the brain under hypoxic stress. Attempts have been made to develop stroke treatments utilizing the neuroprotective function of EPO or recombinant EPO, but treatments using these drugs have faced difficulties due to side effects such as excessive red blood cell production or tumor induction.
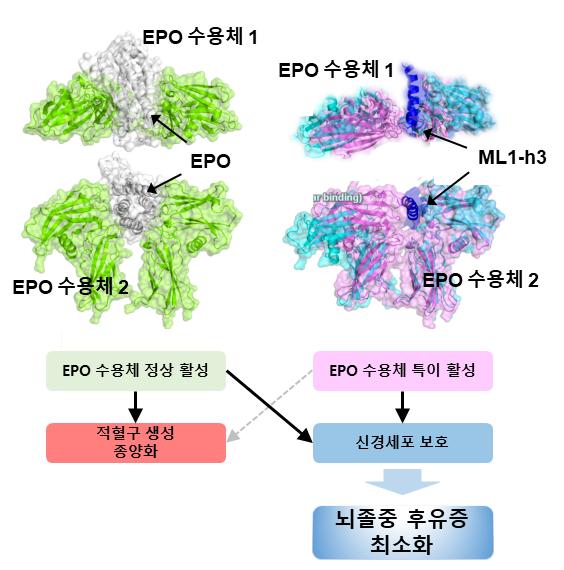
The research team designed new peptide analogs by substituting several amino acids in the helix C, a major region of EPO that binds to the EPO receptor, through molecular modeling to structurally modify it in various ways. The selected peptide analogs were synthesized as drug candidates and demonstrated cell-protective effects. These peptide analogs showed neuroprotective effects against oxidative stress similar to EPO and inhibited reactive oxygen species generation.
Among the peptide analogs, the team focused on ML1-h3, which showed the best cell-protective effect without promoting cell proliferation, as a candidate for stroke treatment. When administered to animals with induced ischemic brain injury, ML1-h3 prevented neuronal cell death and suppressed brain damage to a degree similar to EPO. After one month of long-term administration, unlike EPO, the ML1-h3 peptide did not show hematological toxicity such as excessive red blood cell production.
Many studies have progressed to minimize excessive red blood cell production and tumorigenic effects to use erythropoietin as a treatment for neurological diseases such as stroke. In particular, a ‘first-generation’ erythropoietin peptide therapeutic was proposed by designing only the major binding region to the erythropoietin receptor. In this study, various derivatives were produced using molecular modeling, and among them, the ML1-h3 peptide was developed as a ‘second-generation’ erythropoietin peptide therapeutic with more than 1.5 times enhanced neuroprotective effects compared to conventional erythropoietin, while showing almost no excessive red blood cell production or tumorigenic effects.
For ischemic stroke treatment, it is crucial to prevent brain damage that occurs after urgent reperfusion procedures following cerebral infarction. The ML1-h3 peptide developed in this study is expected to treat such brain damage. Moreover, since existing erythropoietin drugs have demonstrated neuroprotective effects in degenerative neurological diseases such as Alzheimer’s disease, the research team believes ML1-h3 could be used as a more broadly applicable neurological disease treatment.
This study confirmed neuroprotective effects and hematological toxicity under ischemic stress at the animal model level, so simple additional experiments are needed, but it can be considered to have passed the preclinical stage to some extent. Therefore, clinical trials and new drug approval processes are required, which are expected to take about 10 years at the longest. There is still a long way to go.
Peptide drugs have weaknesses in terms of absorption and drug stability in the body. Recently, various chemical technologies have been proposed to overcome these disadvantages of peptide drugs, and using these technologies, it is thought that the practical application of next-generation erythropoietin peptides can be accelerated.
Professor Moon Jae-il said, “This study presents an approach to separate and regulate various functions of biological hormones through specific structural modifications of functional regions based on understanding the activation mechanism of biological hormones, enabling the development of stroke treatments excluding the side effects of erythropoietin drugs,” adding, “It is highly meaningful in that it suggests the direction and possibility of developing many biological protein hormones, which were previously avoided in drug development or patient use due to side effects, into new drugs for patient treatment.”
The results of this study were published in the international journal ‘Redox Biology’ last February.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)