Stabilizing Domestic Supply Difficult with Only Overseas Developed Vaccines
Only One Company Conducting Clinical Trials for 'Popular' mRNA Vaccine
[Asia Economy Reporters Jihee Kim and Chunhee Lee] The reason Celltrion declared its intention to develop a COVID-19 vaccine directly lies in its determination to accelerate the development of a ‘native’ vaccine in South Korea. Amid the global pandemic with the spread of variant viruses, it has become difficult to stabilize domestic vaccine supply solely with overseas-developed COVID-19 vaccines.
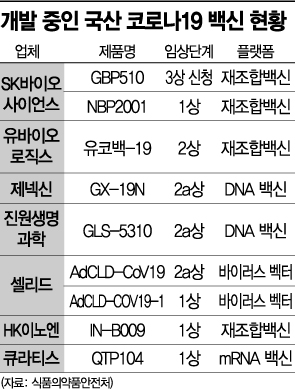
According to the Ministry of Food and Drug Safety on the 4th, there are currently a total of 10 domestic vaccines undergoing clinical trials after receiving approval for clinical trial plans in South Korea. These include five recombinant vaccines, three DNA vaccines, one viral vector vaccine, and one mRNA vaccine. At present, most are still in phases 1 and 2 of clinical trials, so the earliest full-scale launch can be expected around the first half of next year.
The fastest development is attributed to SK Bioscience. Currently developing two candidate substances, ‘GBP510’ and ‘NBP2001,’ SK Bioscience submitted a clinical phase 3 trial plan for GBP510 to the Ministry of Food and Drug Safety in June and is awaiting approval. Once clinical approval is granted, they plan to sequentially apply for phase 3 trials in Europe, Southeast Asia, and other regions. The goal is commercialization in the first half of next year.
Genexine, developing ‘GX-19N’ based on a DNA vaccine platform, is also drawing attention. Genexine, which was the first in South Korea to enter phase 2, received approval last month from Indonesia’s Food and Drug Monitoring Agency (BPOM) for phase 2 and 3 clinical trial plans. An industry insider explained, "There were concerns about the market size as domestic vaccine commercialization is expected only next year, but the recent atmosphere is gradually becoming more hopeful," adding, "Most latecomer domestic companies are continuing development aimed at responding to variant viruses."
However, Curatis is the only domestic company currently conducting clinical trials for the globally popular mRNA vaccines. To develop mRNA vaccines, three major pharmaceutical companies have joined forces. Hanmi Pharmaceutical, GC Green Cross, and ST Pharm recently launched the ‘Next-generation mRNA Vaccine Platform Technology Consortium’ under the support of the Korea Innovative Medicines Consortium (KIMCo). Their blueprint includes producing 100 million doses of mRNA-based COVID-19 vaccines capable of one-time nationwide vaccination by next year and producing 1 billion doses by 2023 for export.
Celltrion is reported to have been preparing in advance for the possibility of entering vaccine development. Seo Jung-jin, Honorary Chairman of Celltrion, said in February, "If vaccine technology independence is not achieved, we are prepared to enter the vaccine field," but added cautiously, "It means we will do it only if there is no other alternative." Regarding this, a Celltrion official stated, "Through this collaboration, we will secure our own mRNA platform technology and apply it to the development of new drugs in various fields to expand our new drug portfolio."
The government views securing domestic vaccines as important for the government’s negotiating power as well. Son Young-rae, Head of the Social Strategy Division at the Central Disaster and Safety Countermeasures Headquarters, said, "It is very important to have domestic vaccines in any form," adding, "Considering stable supply, price, and negotiating power, ultimately, a domestic mRNA vaccine is necessary."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.











