13.21 Million Received First Dose... Vaccination Rate at 25.7%
With Accelerating Vaccination Pace,
Concerns Over Temporary Demand and Supply Mismatch
 On the 16th, at the COVID-19 vaccination center in Seodaemun-gu, Seoul, police forces are assisting with vaccinations as the first dose vaccination target of 13 million people was surpassed, achieving the goal earlier than initially expected. Photo by Jinhyung Kang aymsdream@
On the 16th, at the COVID-19 vaccination center in Seodaemun-gu, Seoul, police forces are assisting with vaccinations as the first dose vaccination target of 13 million people was surpassed, achieving the goal earlier than initially expected. Photo by Jinhyung Kang aymsdream@
[Asia Economy Reporter Lee Chun-hee] As COVID-19 vaccination accelerates and the number of vaccinated individuals surpasses 13 million, attention is focused on whether the government’s goal of achieving ‘herd immunity by November’ will be brought forward. With the Moderna vaccine, the fourth vaccine introduced domestically, beginning distribution on the 16th, the timing of the domestic introduction of the Novavax vaccine, which succeeded in Phase 3 clinical trials, is expected to impact vaccine supply.
The COVID-19 Vaccination Promotion Task Force announced that as of midnight on this day, 13,219,207 people have received their first dose of the COVID-19 vaccine. It took 110 days since vaccinations began on February 26 to surpass 13 million. The corresponding vaccination rate is 25.7%.
In the early stages, it took 39 days to reach 1 million vaccinations, but as vaccine supply increased and vaccination capacity strengthened, it took only 5 days to go from 10 million on the 10th to 13 million. Considering that 1.63 million people have made advance reservations from the 16th to the 19th, it is highly likely that the number will exceed 14 million within this week.
 On the 1st, a transport vehicle carrying 55,000 doses of the Moderna vaccine entered the GC Green Cross Ochang Plant in Ochang-eup, Cheongju-si, Chungbuk. / Photo by Lee Chunhee
On the 1st, a transport vehicle carrying 55,000 doses of the Moderna vaccine entered the GC Green Cross Ochang Plant in Ochang-eup, Cheongju-si, Chungbuk. / Photo by Lee Chunhee
Vaccinations in the second half of the year are expected to proceed smoothly according to the government’s plan. Distribution of the Moderna vaccine began on this day, and the Novavax vaccine, which SK Bioscience is contract-manufacturing (CMO) domestically, demonstrated high efficacy in Phase 3 clinical trials. If these vaccines are supplied domestically in earnest in the second half of the year along with existing vaccines, the government’s target timeline for herd immunity could be accelerated.
The task force plans to complete first-dose vaccinations for 36 million people, 70% of the total population, by September and finish second-dose vaccinations by November to achieve herd immunity. Vaccine imports, which amounted to 19.4 million doses in the first half of the year, are expected to reach 80 million doses in the third quarter and 90 million doses in the fourth quarter.
The Ministry of Food and Drug Safety approved the national release of 55,000 doses of the Moderna vaccine introduced on the 1st on the 15th. Accordingly, excluding 300 doses used for national release approval, 54,700 doses will begin to be administered this week to healthcare workers under 30 years old (including prospective healthcare workers). Currently, the Moderna vaccine is stored at GC Green Cross’s Ochang plant, which is responsible for domestic distribution. A GC Green Cross official explained, "Vaccine shipments are scheduled to begin today." Including this batch, a total of 40 million doses of the Moderna vaccine are planned to be introduced.
Similarly, momentum is expected to build for the introduction of the Novavax vaccine, also planned for 40 million doses. Novavax recently announced that its COVID-19 vaccine showed 90.4% efficacy in Phase 3 clinical trials. In this trial involving about 30,000 participants, the prevention rate against severe infection was 100%, and it showed a 93.2% prevention rate against eight variants, including the ‘Alpha (UK variant)’ and ‘Beta (South Africa variant).’ Novavax plans to apply for emergency use authorization from U.S. and European health authorities within the third quarter.
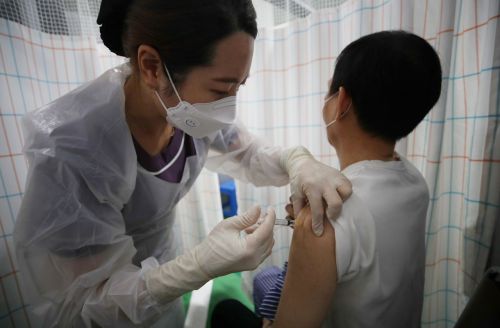 On the 7th, medical staff at a hospital in Dobong-gu, Seoul, are administering the AstraZeneca (AZ) COVID-19 vaccine. [Image source=Yonhap News]
On the 7th, medical staff at a hospital in Dobong-gu, Seoul, are administering the AstraZeneca (AZ) COVID-19 vaccine. [Image source=Yonhap News]
However, as vaccination speed accelerates, temporary mismatches between supply and demand are a concern. According to the task force, first-dose vaccinations with the AstraZeneca (AZ) vaccine currently underway will be temporarily suspended from the 20th. Task Force Director Jung Eun-kyung stated on the 14th that due to differences between vaccine supply and reservations, "It is estimated that about 360,000 excess reservations have occurred among the elderly aged 60 to 74." Many of these individuals are expected to receive vaccinations only next month.
The situation is similar with the Pfizer vaccine. From the 15th, vaccinations began for 200,000 essential workers under 30 years old, healthcare workers, and caregivers who were unable to receive the AZ vaccine in the second quarter due to age restrictions. Although the total target group is 267,000, the number of eligible people significantly exceeded the government’s estimate of about 190,000, resulting in first-come, first-served reservations. The remaining 67,000 are expected to be vaccinated between the 5th and 17th of next month after rebooking from the 28th to the 30th.
As vaccinations proceed rapidly, vaccine supply has not kept pace, creating a reversal phenomenon. Currently, vaccines confirmed for supply in the first half of the year amount to only 835,000 doses of AZ and 1.95 million doses of Pfizer under direct contract (including 650,000 doses introduced on the 16th).
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.

![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)














