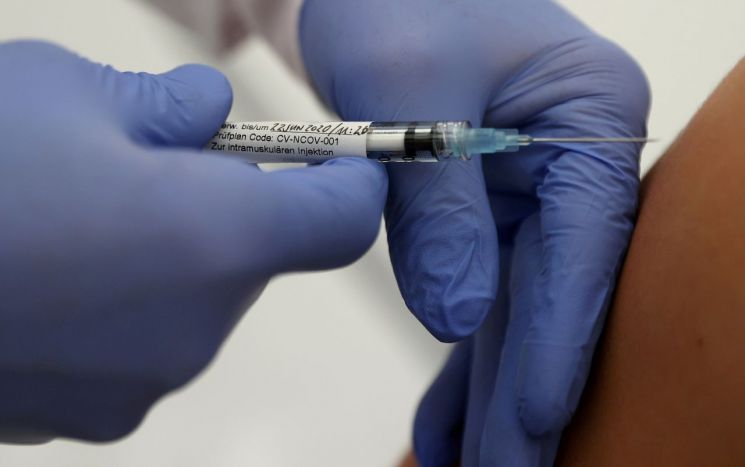
 The CureVac mRNA COVID-19 vaccine is being administered to clinical trial participants in T?bingen, Germany.
The CureVac mRNA COVID-19 vaccine is being administered to clinical trial participants in T?bingen, Germany. [Image source=Reuters Yonhap News]
[Asia Economy Reporter Lee Chun-hee] As the launch of CureVac's messenger ribonucleic acid (mRNA) COVID-19 vaccine from Germany approaches, attention is also focusing on the development of domestic mRNA vaccines.
Currently, COVID-19 vaccines developed or under development domestically and internationally are categorized into messenger ribonucleic acid (mRNA), viral vector, DNA, and synthetic antigen (recombinant) vaccines. Among foreign vaccines, Pfizer and Moderna use mRNA, AstraZeneca (AZ) and Janssen use viral vectors, Inovio uses DNA vaccines, and Novavax uses synthetic antigen vaccines.
Among these, the messenger ribonucleic acid (mRNA) vaccine is a genetic vaccine that uses mRNA to induce an immune response against the COVID-19 virus. Unlike viral vector vaccines such as AZ or Janssen, which have recently been highlighted due to rare blood clot controversies, mRNA vaccines do not use other vectors and have relatively fewer reported side effects. This is why there is a considerable response domestically that says, "I don't want to get AZ, but I can safely get Pfizer." Additionally, since it can be made by synthesizing only specific genes, the development of candidate substances is fast, and it is highly regarded for its potential use against variant viruses as well as other diseases.
This is why there is keen interest in the Phase 3 clinical trial results of the mRNA COVID-19 vaccine from CureVac, which is expected to be announced soon following Pfizer and Moderna.
In particular, CureVac's vaccine is attracting attention because, unlike Pfizer and Moderna vaccines that require ultra-cold chain distribution, it can be distributed in regular refrigeration facilities. Currently, domestically, unlike the Pfizer vaccine, which is administered only at limited vaccination centers for this reason, CureVac's vaccine can be administered at general hospitals like the AZ vaccine. The vaccination schedule is developed as two doses, similar to Pfizer and Moderna.
Currently, CureVac has requested approval for use in the European Union (EU) and has signed supply contracts for 405 million doses with EU countries. CureVac plans to produce 300 million doses this year and up to 1 billion doses next year.
Before CureVac, BioNTech, which co-developed the first mRNA COVID-19 vaccine known as the Pfizer vaccine, is also a German pharmaceutical company. This is also cited as a reason why Germany has taken a firm stance against the recent discussions on the temporary waiver of intellectual property rights (IPR) for COVID-19 vaccines. The German government spokesperson emphasized on the 6th, "We support the goal of supplying COVID-19 vaccines worldwide, but the bottleneck in vaccine production is not patent rights but production capacity and quality standards," adding, "Protection of intellectual property rights is the source of innovation."
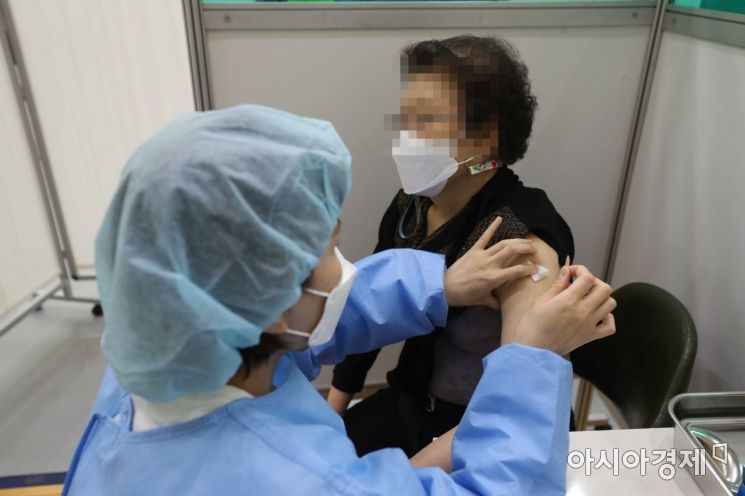 Elderly people aged 75 and over are receiving the Pfizer vaccine at the Jung-gu COVID-19 Vaccination Center set up at the Chungmu Sports Center in Jung-gu, Seoul. Photo by Mun Ho-nam munonam@
Elderly people aged 75 and over are receiving the Pfizer vaccine at the Jung-gu COVID-19 Vaccination Center set up at the Chungmu Sports Center in Jung-gu, Seoul. Photo by Mun Ho-nam munonam@
Domestically, the development of mRNA COVID-19 vaccines is still uncertain. Currently, five companies?SK Bioscience, Genexine, GeneOne Life Science, Cellid, and UbioLogics?have received approval for vaccine clinical trial plans and are conducting trials, but all are developing vaccines of types other than mRNA. SK Bioscience and UbioLogics are developing synthetic antigen vaccines, Genexine and GeneOne Life Science are developing DNA vaccines, and Cellid is developing an adenovirus vaccine.
The quarantine authorities plan to accelerate the development of domestic mRNA vaccines. The goal is to start clinical trials for mRNA vaccines this year.
On the 7th, Kwon Jun-wook, Deputy Director of the Central Disease Control Headquarters (CDCH) at the Korea Disease Control and Prevention Agency (KDCA) and Director of the National Institute of Health, said at a briefing, "The mRNA vaccine platform is a technology that can be expanded to other chronic diseases beyond infectious diseases such as cancer, so it must be secured," adding, "The goal is to start clinical trials for RNA vaccine technology within this year."
Deputy Director Kwon said, "We are not the world’s best in vaccine development," and added, "There has been a lack of investment, interest, support, and effort, and the accumulated time has been short." However, he expressed determination, saying, "COVID-19 has been a turning point. We inevitably have to develop the mRNA vaccine platform, and because it is urgent, I believe it will be developed, and we are putting all our efforts into it, so it will succeed."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)















