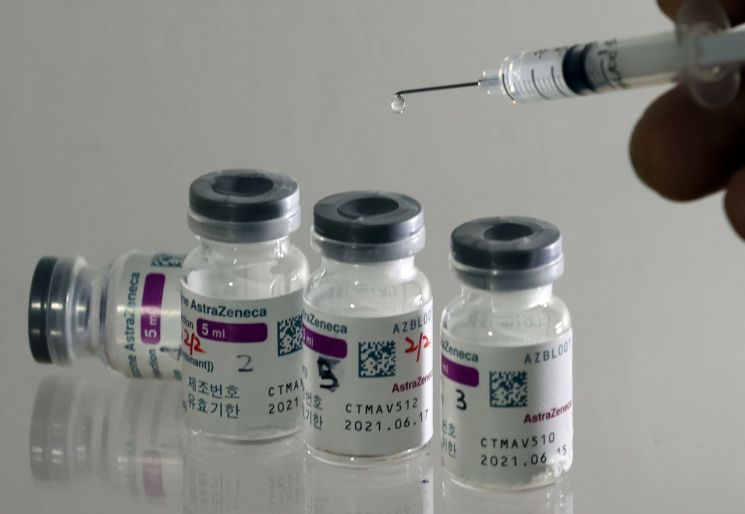
 On the 17th, AstraZeneca's COVID-19 vaccine became the first domestically introduced COVID-19 vaccine to receive national release approval.
On the 17th, AstraZeneca's COVID-19 vaccine became the first domestically introduced COVID-19 vaccine to receive national release approval. [Image source=Yonhap News]
[Asia Economy Reporter Lee Chun-hee] The approval process for COVID-19 vaccines is expected to accelerate as national batch release approval will be possible before the product license review.
The Ministry of Food and Drug Safety (MFDS) announced on the 19th that it has issued an administrative notice for partial amendments to the "Regulations on Designation, Approval Procedures, and Methods of National Batch Release Approval" containing these details.
Biological products such as vaccines must undergo a national batch release approval process to verify quality once more through testing and data review before sales, even after product licensing. The MFDS has promoted this amendment to streamline the rapid national batch release approval procedure for vaccines in pandemic situations such as the emergence of new infectious diseases.
First, the amendment allows products requiring rapid release, such as those related to public health and infectious disease pandemics, to apply for batch release approval even before completing the product license procedure. Previously, national batch release approval could only be applied for after the review of standards and test methods was completed. The MFDS expects that this will enable swift quality verification of vaccines for infectious disease prevention and timely supply of necessary quantities.
Additionally, the timing for applying risk assessment results has been clearly defined as "from April 1 to March 31 of the following year." Risk assessment is a system that sets risk levels for each product based on the previous year's biological product batch release approval history and results, determining the inspection items and methods for the relevant year. The MFDS prepared this amendment to enhance predictability in national batch release approval.
The national batch release approval procedures and submission requirements for vaccines preventing bioterrorism and pandemic infectious diseases will also be improved. To establish specific procedures for rapid batch release approval, a new process has been introduced allowing rapid national batch release approval upon request by the head of relevant ministries for public health or national defense purposes. Submission materials can also be separately determined according to the situation, enabling flexible responses during pandemic conditions.
The MFDS stated, "We will continue to closely monitor the COVID-19 situation and operate a rational and flexible national batch release approval system to ensure that the public can receive safe vaccines promptly, making every effort in system management."
The MFDS will accept opinions on this notice until the 11th of next month. Detailed administrative notice information is available on the MFDS website.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Clutching a Stolen Dior Bag, Saying "I Hate Being Poor but Real"... The Grotesque Con of a "Human Knockoff" [Slate]](https://cwcontent.asiae.co.kr/asiaresize/183/2026021902243444107_1771435474.jpg)