Chong Kun Dang's 'Lucenbace' Domestic Approval
Second Own Biosimilar After 'Nesvel'
Traditional Pharma Companies Also Developing and Launching
Dong-A ST 'Darbepoetin Alfa' 'DMB-3115'
LG Chem 'Yusept' 'Adalimumab BS MA'
Mostly Limited to Asia Including Japan, Regrettable
Dong-A ST and Samchundang Undergoing FDA-Approved Clinical Trials
[Asia Economy Reporter Lee Chun-hee] Traditional pharmaceutical companies are increasingly challenging the Korean biosimilar (biopharmaceutical generic) industry, which has been represented by bio companies such as Celltrion and Samsung Bioepis. While continuously succeeding in launching biosimilars for key biopharmaceuticals, biosimilars directly targeting approval from the U.S. Food and Drug Administration (FDA) are also being developed.
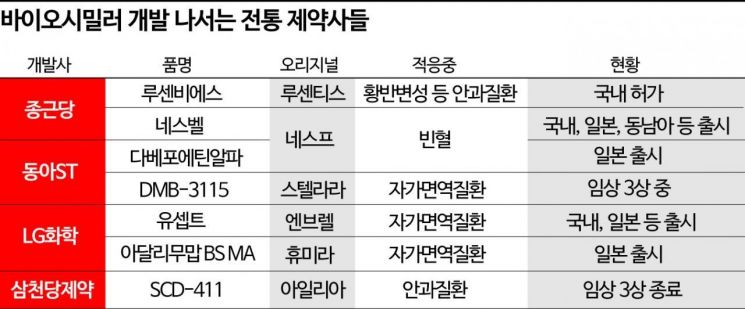
Recently, Chong Kun Dang Pharmaceutical received domestic approval for 'Lucentis BS,' a biosimilar of 'Lucentis,' a treatment for ophthalmic diseases such as wet age-related macular degeneration. Notably, it succeeded in obtaining approval for all full-label indications approved for Lucentis in Korea, including wet age-related macular degeneration, diabetic macular edema, and diabetic retinopathy. Chong Kun Dang plans to launch the product soon after completing the National Health Insurance registration process. With this, Lucentis BS became Chong Kun Dang's second self-developed biosimilar following 'Nesvel,' launched in 2019. Nesvel, a biosimilar of the anemia treatment 'Nesp,' has expanded not only domestically but also to Japan, Southeast Asia, and the Middle East.
Seizing the Growing Biosimilar Market... Focus on the Asian Market
In addition to Chong Kun Dang, traditional pharmaceutical companies such as Dong-A ST, LG Chem, and Samchundang Pharmaceutical are actively pioneering the biosimilar market. Dong-A ST is also selling 'Darbepoetin Alfa,' a biosimilar of Nesp, in Japan. After completing Phase 1 clinical trials, it transferred development and sales rights within Japan to Sanwa Kagaku Kenkyusho (SKK). Last year, sales exceeded 10 billion KRW annually.
LG Chem has developed and is selling 'Yusept' and 'Adalimumab BS MA,' biosimilars of the autoimmune disease treatments 'Enbrel' and 'Humira.' LG Chem began developing Yusept in 2010, before the biosimilar market was established, and launched it in Japan and Korea in 2018. Notably, it was the first Enbrel biosimilar to successfully enter the Japanese market, achieving a 40% market share of the 'Etanercept' ingredient as of the first quarter. Adalimumab BS MA was also launched in Japan last year, aiming to expand its market.
However, it is still difficult to say that traditional pharmaceutical companies are actively entering the biosimilar market. Most product launches are still concentrated in Asia, including domestic, Japan, Southeast Asia, and the Middle East. The only Korean biosimilars approved in the world's largest pharmaceutical market, the United States, remain those made by Celltrion ('Remsima,' 'Truxima,' 'Herzuma,' 'Vegzelma') and Samsung Bioepis ('Eticovo,' 'Renflexis,' 'Hadlima,' 'Ontruzant,' 'Biouviz'). Therefore, there are calls to closely watch whether companies are targeting the U.S. market from the clinical stage.
Samchundang Pharmaceutical recently completed Phase 3 clinical trials for 'SCD-411,' a biosimilar of 'Eylea,' a treatment for ophthalmic diseases. After receiving Investigational New Drug (IND) approval from the FDA, the clinical trials were conducted in 15 countries, including the U.S. and Japan, involving 576 patients with macular degeneration. Samchundang plans to submit approval applications in Japan, the U.S., Korea, and Europe after receiving the final Phase 3 clinical trial report around January next year.
Dong-A ST is also conducting Phase 3 clinical trials for 'DMB-3115,' a biosimilar of 'Stelara,' an autoimmune disease treatment whose patent expires next year, including in the U.S. Outside the U.S., recruitment of 605 patients with plaque psoriasis in European countries such as Poland and Estonia was completed at the end of last year, and the trials are expected to be completed within this year.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.